INTRODUCTION
Owing to the increase in life expectancy, increasing numbers of older patients require hospital care and polymedication. Recently introduced, oral anticoagulants are currently one of the most widely administered pharmacological medications.1)
Oral anticoagulants are widely administered as therapeutic options for atrial fibrillation (AF), thromboembolic disease prevention and treatment (especially in the postoperative period), acute coronary syndrome, and heparin-induced thrombopenia.2,3) These agents act in the coagulation cascade as direct thrombin inhibitors administered parenterally (bivalirudin or argatroban) or orally (dabigatran) or as direct factor Xa inhibitors administered orally (rivaroxaban, apixaban, edoxaban, and betrixaban). In addition to showing clinical superiority over other anticoagulants, these drugs offer advantages for patients, including the need for fewer controls to adjust the dose and less interaction with food, among others, which has led them to replace classic anticoagulants such as warfarin and acenocoumarol.
CLINICAL CASE
An 89-year-old woman with a history of hypertension, dyslipidemia, and AF was treated with bisoprolol (2.5 mg), apixaban (2.5 mg), ramipril (2.5 mg), and simvastatin (10 mg).
She presented for a consultation because of the spontaneous appearance of purpuric macules in both lower extremities, which had evolved over approximately 15 days. These macules were not pruritic and the patient had no fever, proteinuria, hematuria, or additional symptoms. However, she had started anticoagulant therapy with apixaban 1 month before the diagnosis of paroxysmal AF.
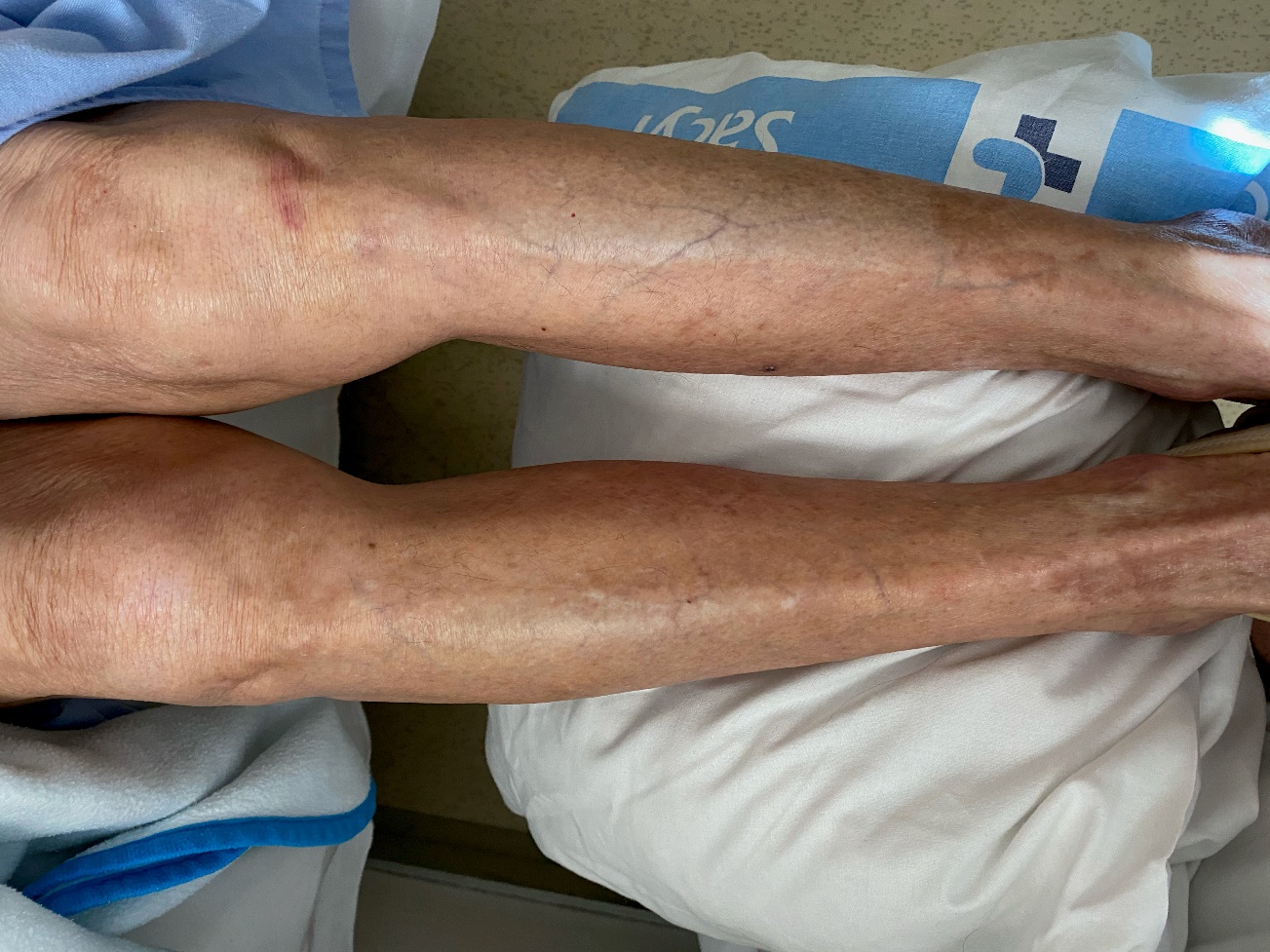
Physical examination (Fig. 1) revealed confluent symmetric petechial lesions in both lower limbs from the dorsal aspect of the foot to the thigh. The lesions were not raised and did not whiten with in vitro pressure. No other abnormalities were noted. An electrocardiogram (ECG) showed AF at 90 bpm, whereas posteroanterior chest radiography showed no alterations.
Laboratory examination revealed a leukocyte count of 11.1 × 103/µL (12.5% neutrophils), red series, and normal platelet count. Her erythrocyte sedimentation rate was 34 mm/hr, and she showed normal coagulation (plasmin inhibitor [PI], 73%; international normalized ratio (INR), 1.32; activated partial thromboplastin time [APTT], 29.4 seconds), except for fibrinogen levels (560 mg/dL). Biochemical analysis showed normal ion levels and renal function (glutamate pyruvate transaminase [GPT], 23 U/L; total bilirubin [BT], 0.64 mg/dL; gamma-glutamyl transferase [GGT], 186 U/L; alkaline phosphatase [FA], 193 U/L; lactic acid dehydrogenase [LDH], 234 U/L; C-reactive protein [CRP], 28 mg/L). The patient showed a normal lipid profile as well as normal venous blood gas and complement component 3 and 4 levels. She also showed negative results for antinuclear antibodies, anti-Ro, anti-La, c-ANCA, and p-ANCA. Further, serological tests yielded negative results for hepatitis B, hepatitis C, human immunodeficiency virus (HIV), varicella-zoster virus, and infectious mononucleosis. Finally, results of the QuantiFERON test were negative as well.
Given our clinical suspicion of a drug side effect, the Naranjo algorithm was applied, and the patient’s score was 6, which suggested that an adverse drug had occurred. We suspended the administration of the potentially causative drug, apixaban; performed a skin biopsy; and commenced treatment with low-molecular-weight heparin. The biopsy findings were comparable to those for vasculitis; therefore, a topical corticosteroid was prescribed. After 3 weeks without the drug and with topical corticosteroid treatment, the lesions disappeared (Fig. 2). Furthermore, the altered liver function parameters observed at admission, which were possibly caused by hepatic metabolism of apixaban, normalized. A definitive diagnosis was made on the basis of the results of the biopsy and the clinical improvement after drug withdrawal.
DISCUSSION
Assessment of the risks and benefits of treatments for patients is standard process in clinical practice. This involves identifying the best therapeutic option for patients that would result in the fewest side effects. This goal is paradigmatic in the case of anticoagulant therapies, which seek to avoid thrombus formation, with the lowest possible risk for side effects, especially bleeding. New oral anticoagulants show reduced risks for thrombotic events; however, they are not without side effects. Cutaneous side effects are rare, occurring in fewer than 1/100 according to the technical data sheet (apixaban).4) However, drug interactions must be ruled out by review of basic medication in polymedicated patients such as our patient.
Cutaneous small-vessel vasculitis (CSVV) has been reported in a very few cases after exposure to direct oral anticoagulants (DOACs). A recent review of the literature reported no more than 50 cases; among these cases, 50% reported a time to onset of within 10 days of DOAC exposure. When specified, the predominant type of CSVV reported was leukocytoclastic vasculitis (n=31).5)
The 2012 Chapel Hill International Consensus Conference grouped cutaneous arteritis, primary central nervous system vasculitis, isolated aortitis, and cutaneous leukocytoclastic vasculitis under the term single-organ vasculitis. This entity is associated with a wide spectrum of systemic conditions, neoplasms, infections, and drug hypersensitivities.6) Histologically, these cases are classified as small-vessel vasculitis (<50 μm in diameter). Localized or systemic damage occurs because of complement action and the release of pro-inflammatory cytokines via the activation of immune complexes, through antigen–antibody interactions in the vascular wall, or by leukocyte activation of ANCA.7)
We propose that the most appropriate therapy for each patient should be determined on the basis of a series of indicators, namely, the severity of skin involvement, the presence of extracutaneous manifestations, and the evolution time. Treatments may vary from symptomatic treatment to the use of colchicine or corticosteroids.8,9) In our patient, drug withdrawal and topical treatment were sufficient.