 |
 |
- Search
| Ann Geriatr Med Res > Volume 25(4); 2021 > Article |
|
Abstract
Background
We investigated the comorbidities of individuals who were prescribed statins to identify the use of bone mineral density (BMD)-reducing drugs, examine polydrug use trends involving these drugs, and explore their relationship with osteoporosis.
Methods
We analyzed claims data from the Korean National Health Insurance Service (January 2014–December 2018). We sampled 20% of 8,379,419 patients aged ≥50 years who were prescribed statins. Among them, we analyzed the data of those who were administered two or more prescriptions for 14 days or longer within 6 months of the initial date of statin prescription. Data on comorbidities and drugs that can potentially reduce BMD were obtained. Osteoporosis-related diagnoses were obtained as an outcome measure. The relationship between statins and BMD-reducing drugs was analyzed using logistic regression.
Results
Among the 4,138 statin users aged 50 years or older, 552 were diagnosed with osteoporosis. The most common comorbidity in statin users was hypertension, followed by ischemic heart disease, diabetes mellitus, and stroke. The most frequently administered BMD-reducing drugs were proton pump inhibitors (PPIs). The osteoporosis diagnosis rate was higher in patients who were prescribed both statins and PPIs or both statins and levothyroxine than in those using only a statin.
The proportion of older adults in South Korea is increasing, including that of individuals aged ≥75 years who often have complex health problems and show a functional decline. The older adult population is predicted to account for more than half of the total population by 2038.1) The incidence of “polydrug use” defined as the use of more than one drug or multiple types of drugs at the same time or sequentially,2) is rising owing to extended life expectancy and the increasing occurrence of complex chronic diseases in the older adult population.3-7) Polydrug use can increase the risk of the prescription of potentially inappropriate medications (PIM) in older adults. A previous study that assessed the rate of PIM among Korean older adults observed the prescription of at least one PIM in 80.96% of older patients and that polydrug use was a significant risk factor for PIM prescription.8)
The National Institutes of Health designated osteoporosis as a skeletal disorder with an elevated risk of fracture owing to weakening bone strength.9) The 2008–2011 Korea National Health and Nutrition Examination Survey (KNHANES) reported a prevalence of osteoporosis among individuals aged 50 years or older of 22.4%. The prevalence in women (37.3%) is approximately five-fold higher than that in men (7.5%). Moreover, the incidence of osteoporosis increases with age with 68.5% of women aged 70 years or older experiencing osteoporosis.10)
Dyslipidemia, a major risk factor for cardiovascular diseases, is a chronic disease that requires continuous management in older adults. Furthermore, an analysis of the 2002–2018 National Health Insurance Service (NHIS) dataset published in the Dyslipidemia Fact Sheet in Korea in 202011) showed that 7.69 million people were prescribed medications for dyslipidemia. A recent study conducted outside of Korea reported that the risk of osteoporosis increases with the dosage of statin, a drug used to treat dyslipidemia.12) This finding called for an evaluation of the use of drugs that reduce bone mineral density (BMD) among statin users.
The use of proton pump inhibitors (PPIs) is increasing owing to the recent increasing prevalence of digestive diseases in patients with obesity and in older adults.13) Studies have reported adverse events such as reduced BMD and osteoporosis caused by the prolonged use of PPIs.14) A classic example of a potassium-competitive acid blocker is revaprazan. Although revaprazan has been gaining popularity as a medication for gastroesophageal reflux disease, patients often take other drugs for metabolic disorders, including non-steroidal anti-inflammatory drugs. Therefore, studies on the resultant adverse drug reactions are required.15)
Tenofovir is commonly used to treat hepatitis B and human immunodeficiency virus infections. It reportedly induces Fanconi syndrome and ultimately leads to reduced BMD, osteomalacia, and electrolyte imbalance.16) A retrospective cohort study reported that the risk of osteoporosis fracture increased by 12% with tenofovir use.17)
Levothyroxine is considered the standard of care for hypothyroidism and, thus, is widely prescribed. However, excessive levothyroxine use accelerates bone loss and may have deleterious effects, such as osteoporosis. This calls for its cautious prescription, particularly to older adults and postmenopausal women.18)
The recently introduced sodium-glucose co-transporter 2 (SGLT2) inhibitors are hypoglycemic agents involved in glucose resorption in the kidney.19) However, SGLT2 inhibitor users have been shown to result in significantly reduced hip BMD.20)
Therefore, we examined comorbidities in statin users and identified drugs that might reduce BMD. We also explored trends in polydrug use involving these drugs and investigated their relationship with osteoporosis.
As the study subjects were older adults taking statins, “stain users” were defined based on previous cohort studies21,22) to extract as many users as possible. We enrolled patients prescribed any type of statin and with more than two prescriptions lasting 14 days or longer within 6 months of the initial statin prescription date (index date) between January 2014 and December 2018. The patients’ statin prescription history was determined using the active ingredient codes for each statin type. The following statins were analyzed: atorvastatin, rosuvastatin, lovastatin, simvastatin, pravastatin, fluvastatin, and pitavastatin. The total days of statin supplies were calculated based on the number of days in the prescription.
This retrospective cohort study used NHIS claims data and enrolled a total of 8,379,419 patients aged 50 years or older who were prescribed statins between January 2014 and December 2018. Of these patients, we sampled 20% (n=1,675,884). After excluding patients aged under 50 years and 90 years and older (n=16,319) based on the index date, 1,659,565 patients remained. Among them, we selected 1,385,035 patients who had been prescribed statins at least twice for 14 days or longer within 6 months of the index date. The additional exclusion criteria were (1) patients prescribed statins within 2 years before the index date; (2) patients diagnosed with dyslipidemia (I78.0–I78.9), osteoporosis (M81 and M82), or osteoporotic fracture (M80, S72.0, S72.1, S22.0, S22.1, S32.0, M48.4, M48.5, S52.5, and S52.6) within 2 years before the index date; (3) patients who had taken osteoporosis medication (including calcium and vitamin D) within 2 years before the index date; (4) patients prescribed dyslipidemia medications other than statins within 2 years before the index date; and (5) patients whose statin dose was adjusted during statin therapy. Both main and sub-diagnoses were checked (Fig. 1).
We included sex and age as demographic factors in the analysis of osteoporosis incidence according to statin use. The analyzed comorbidities were rheumatoid arthritis (M06); hypertension (I10–I15); ischemic heart diseases (I20–I25); diseases of arteries, arterioles, and capillaries (I70–I79); stroke (I63, I64); diabetes (E10, E11); chronic renal insufficiency (N17–N19); nicotine dependency (F17); overweight and obesity (E65–E68), chronic obstructive pulmonary disease (J44); asthma (J45); and Crohn’s disease (K50). We applied the Korean Standard Classification of Diseases codes, which follow the 10th revision of the International Statistical Classification of Diseases (ICD-10). We checked for cases in which these diagnosis codes appeared at least once between the index date and final date, regardless of whether it was a main diagnosis or sub-diagnosis. Among the drugs that potentially reduce BMD (PPIs, revaprazan, tenofovir, lithium, levothyroxine, and prednisone), oral (sulfonylurea, glinide, metformin, alpha-glucosidase inhibitor, thiazolidinedione, DPP-4 inhibitor, and SGLT2 inhibitor) and injectable (insulin and GLP-1 inhibitor) drugs for treating diabetes mellitus (DM) that are associated with a high fracture risk among older adults with dyslipidemia were identified. We selected cases in which these drugs were prescribed for 3 months or longer between the index and final dates.
In this study, the outcome was osteoporosis-related diagnoses. Osteoporosis was defined as the presence of least one of the following codes: M81 (osteoporosis without current pathological fracture), M82 (osteoporosis in diseases classified elsewhere and defined as osteoporosis in multiple myelomatosis or endocrine disorders), M80 (osteoporosis with current pathological fracture), S72.0 or S72.1 (hip fracture), S22.0, S22.1, S32.0, M48.4, or M48.5 (spine fracture), and S52.5 or S52.6 (distal radius fracture).
The maximum follow-up period was 2 years. Follow-up was terminated if a patient was diagnosed with osteoporosis or died. Furthermore, because the data only covered health care utilization until December 31, 2018, all follow-up was terminated on this date. We set the maximum follow-up period to 2 years to eliminate potential confounding effects, such as the passage of time, that may have contributed to the onset of osteoporosis.
This study was exempted for review by the Institutional Review Board at Seoul Medical Center (No. SEOUL 2020-01-004). Moreover, the data were obtained from the NHIS and were accessed in the analysis center.
We analyzed the participants’ general characteristics, comorbidities, and use of BMD-reducing drugs using descriptive statistics. The frequencies of comorbidities and BMD-reducing drug use were analyzed by sex, age, and statin type. We also analyzed the frequencies of patients undergoing bone density testing at the time of, or within 1 year of, osteoporosis diagnosis. The frequency of statin supply was analyzed by statin type; we also determined the frequency of use of each statin type according to comorbidity, as well as the frequency of polydrug use with BMD-reducing drugs by statin type. We then applied logistical regression to examine polydrug use trends in patients prescribed BMD-reducing drugs according to the prescribed statin type who were also diagnosed with osteoporosis. All statistical analyses were performed using SAS version 9.4 (SAS Institute Inc., Cary, NC, USA) software with a statistical significance level set to 0.05.
A total of 4,138 patients aged 50 years or older were prescribed statins between January 2014 and December 2018. Among these, 2,567 were male (62%) and 1,571 were female (38%), including 1,326 males aged 50–64 years (M50–64, 32.0%), 1,241 males aged 65–89 years (M65–89, 30.0%), 743 females aged 50–64 years (F50–64, 18.0%), and 828 females aged 65–89 years (F65–89, 20.0%). Statins were prescribed for 441.2, 427.4, 338.9, and 384.4 days in the M50–64, M65–89, F50–64, and F65–89 groups, respectively. The mean number of days of statin use among statin users was 441.2. A total of 552 patients were diagnosed with osteoporosis: 47 (8.5%) M50–64, 149 (27%) M65–89, 106 (19.2%) F50–64, and 250 (45.3%) F65–89. Seventy-five of these patients were advised to undergo dual-energy X-ray absorptiometry (DXA) within 1 year of their osteoporosis diagnosis: 5 (6.7%) M50–64, 19 (25.3%) M65–89, 15 (20.0%) F50–64, and 36 (48.0%) F65–89 (Table 1).
Most statin users showed no comorbidities (n=2,078); among those with comorbidities, the most common was hypertension (n=2,051), followed by ischemic heart disease (n=1,326), DM (n=1,207), and stroke (n=1,183). Among statin types, rosuvastatin was the most frequently prescribed (n=2,538; 61.3%), followed by atorvastatin (n=1,001; 24.2%), and pitavastatin (n=569; 13.8%) (Table 2).
Among patients without comorbidities, the most frequently prescribed statin was rosuvastatin (n=1,279), followed by atorvastatin (n=528) and pitavastatin (n=259). Among all patients with comorbidities, including hypertension, ischemic heart disease, DM, and stroke, the most frequently prescribed statin was rosuvastatin (n=1,259, 812, 737, and 722, respectively), followed by atorvastatin (n=473, 300, 276, and 270, respectively), and pitavastatin (n=310, 209, 190, and 188, respectively) (Fig. 2).
Regarding BMD-reducing drug use among statin users, PPIs were the most frequently prescribed (n=1,734), followed by levothyroxine (n=136), tenofovir (n=12), lithium (n=9), and revaprazan (n=6). None of the patients used prednisone. Oral DM drugs were prescribed to 2,501 patients and injectable DM drugs to 79 cases, whereas 384 patients were not prescribed any additional drugs. Among statin users who were diagnosed with osteoporosis, PPIs were the most frequently prescribed medications (n=321), followed by levothyroxine (n=22), revaprazan (n=3), and tenofovir (n=1). None of these patients were prescribed lithium or prednisone. Oral DM drugs were prescribed to 328 of the diagnosed patients and injectable DM drugs to 11 patients, whereas 34 of the diagnosed patients were not prescribed any additional medications (Fig. 3).
The prevalence of osteoporosis was 1.84 times higher among statin users concomitantly prescribed PPIs than among those who did not take additional drugs. This was particularly evident for pitavastatin (2.40-fold), rosuvastatin (1.83-fold), and atorvastatin users (1.82-fold) concomitantly prescribed PPIs. The prevalence of osteoporosis was also higher among statin users concomitantly prescribed levothyroxine (1.81-fold) than among those who were not prescribed additional drugs, especially among concomitant pitavastatin-levothyroxine users (7.03-fold increase). We observed no significant differences in the prevalence of osteoporosis in patients prescribed revaprazan, tenofovir, oral DM drugs, or injectable DM drugs concomitantly with a statin (Table 3).
In this study, we examined a nationwide sample of patients who were prescribed statins between January 2014 and December 2018 for the treatment of dyslipidemia, a major cause of mortality among older adults and a cardiovascular disease risk factor. We analyzed the rates of osteoporosis diagnosis, the prescription of DXA following diagnosis and treatment, and the use of BMD-reducing drugs among patients diagnosed with osteoporosis in two age groups: 50–64 (at 50 years of age, osteoporosis incidence begins to increase) and 65–89 years.
Statins, which are used to treat dyslipidemia, reduce low-density lipoprotein levels by inhibiting β-hydroxy β-methylglutaryl coenzyme A (HMG-CoA) reductase, the most important enzyme in cholesterol synthesis.23) However, they are associated with adverse events, such as hepatotoxicity, myalgia, hyperglycemia, and chronic renal insufficiency, as well as risks from drug interactions. These effects vary widely across different statin types and clinical dosages.23) A meta-analysis of 33 studies that examined the association between statins and osteoporosis reported that statins lowered the risk of fracture, particularly in men.24) In addition, a study that analyzed data from the NHIS in Taiwan reported that statins lowered the risk of osteoporosis and fracture.25) In an analysis of 350,000 Austrians who had taken statins for at least 1 year and 7.80 million Austrians diagnosed with osteoporosis from 2006 to 2007, the low-dose statin group had a low risk of osteoporosis, whereas the high-dose statin group had a 2.04-fold higher risk of osteoporosis.12) In the present study, we only enrolled patients who had been taking statins to eliminate the effect of statin use or non-use on BMD. The study in Austria12) only assessed the association (trend) of osteoporosis with varying doses of the same statin, without analyzing the rate of osteoporosis in relation to different intensities of statin use (high, moderate, or low). Hence, subsequent studies are needed to assess the associations between osteoporosis and statin type and intensity.
The results of previous studies on the associations between PPI use and osteoporosis are inconsistent;26,27) thus, the specific durations, doses, and use of PPIs that elevate the risk of fracture remain unknown. A previous study that analyzed health insurance claims data reported an increased risk of osteoporotic fracture with PPIs use in women who underwent their health check-ups at a transitional life phase at the age of 66 years. The odds ratio (OR) for fractures during the follow-up period was 1.13 for individuals who took PPIs compared to that in individuals who took histamine-2 receptor antagonists (H2RAs) and the OR for hip fracture was even higher (1.51). Moreover, the risk of fracture increased with the duration of PPI use during follow-up.28) In our study, the OR for osteoporosis was 1.84 among statin users with concomitant PPI use compared with that in those who did not take any other prescribed drugs and was 2.40 among pitavastatin users, 1.82 among atorvastatin users, and 1.83 among rosuvastatin users. Furthermore, the OR for osteoporosis was 1.81 among statin users concomitantly prescribed levothyroxine and increased to 7.03 among those concomitantly prescribed pitavastatin and levothyroxine. Hence, bone density testing should be performed in patients prescribed these drugs. The tests would fall within the insurance coverage criteria (disease or use of drugs that may induce osteoporosis).
Among DM drugs, the use of SGLT2 inhibitors reduced hip BMD,20) whereas canagliflozin has been associated with increased fracture risk.29) In contrast, a meta-analysis of the risk of fracture linked to SGLT2 inhibitors in patients with DM reported fracture incidence rates of 1.59% and 1.56% in the SGLT2 inhibitor and control groups, respectively. Compared to a placebo, canagliflozin (OR=1.15; 95% confidence interval [CI], 0.71–1.88), dapagliflozin (OR=0.68; 95% CI, 0.37–1.25), and empagliflozin (OR=0.93; 95% CI, 0.74–1.18) did not significantly increase the risk of fracture.30) In our study, the prevalence of osteoporosis was not significantly higher among statin users who concomitantly took oral DM drugs. However, further studies are needed, as we did not conduct separate analyses according to the composition of the oral DM drugs.
This study differs from other studies in that it compared the prevalence of osteoporosis in patients taking a combination of statin—a hyperlipidemia drug used to lower the risk of cardiovascular and cerebrovascular disease that accounts for a large proportion of comorbidities osteoporosis—and drugs known to reduce BMD. We found that the risk of osteoporosis was higher in patients prescribed both statins and PPIs and those who received both statins and levothyroxine compared to those who took other drugs. Therefore, bone density tests should be performed. Moreover, when prescribing drugs that induce bone density reduction, alternative prescriptions should be considered. Regular bone density tests are also needed for patients diagnosed with osteoporosis or who have risk factors for bone density reduction. Furthermore, a study on the possible interactions of PPIs and levothyroxine with statins is needed. Our study results showed the negative effects of polydrug use by focusing on multiple drugs. Older patients are often prescribed a variety of drugs by different doctors and also take non-prescription drugs, various herbal medicines, and health supplements. The many cases of polydrug use underscore the need for a thorough monitoring system and caution to reduce the unnecessary use of drugs that may interact in older patients.
The number of patients was limited because this study only assessed older patients taking statins and it was difficult to apply the medication possession ratio (MPR) or the 12-month cumulative persistence rate relative to the persistence of dosing. To exclude the characteristics of the older adults and focus on drug interactions, the size of the population must be increased by including younger generations and ensuring the continuity of sufficient drug intake. In addition, a broader patient group rather than just those taking statins may be required to investigate the complex relationships among various drugs. In addition, additional studies on the effect of statin intensity on bone density are needed.
ACKNOWLEDGMENTS
FUNDING
This study was supported grants by the Korea Geriatrics Society. The National Health Information Database was provided by the National Health Insurance Service of Korea.
AUTHOR CONTRIBUTION
Conceptualization, KSK, HJH, KBK, JCH, KMY, AJH; Data curation, JCH, KMY, KDR, PYH, KBK; Funding acquisition, KSK; Investigation, HJH; Methodology, HJH, KBK, JCH, KMY, KDR, PYH,AJH; Project administration, KSK, HJH, KBK; Supervision, KSK; Writing-original draft, HJH, KBK, KSK; Writing-review & editing, HJH, KBK, JCH, KMY, KDR, PYH.
Fig. 3.
Bone mineral density-reducing drug use among statin users and statin users with osteoporosis.
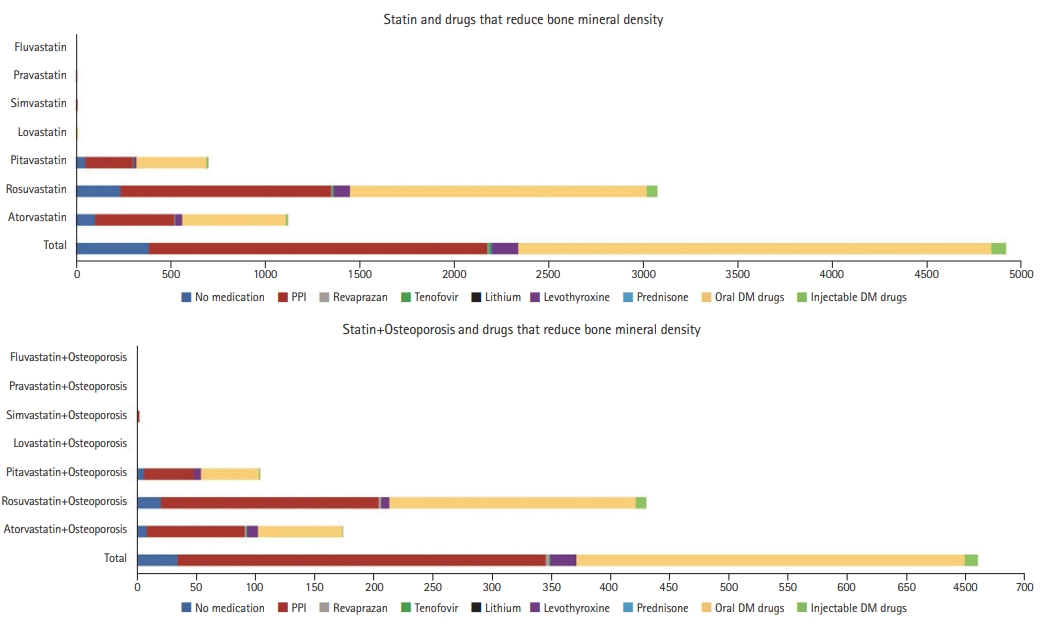
Table 1.
General characteristics of statin users by sex and age group (n=4,138)
Table 2.
Analysis of comorbidities and statin type (n=4,138)
Table 3.
Relationship between osteoporosis and bone mineral density-reducing drug use among statin users
REFERENCES
1. Statistics Korea. Elderly population statistics 2020 [Internet]. Daejeon, Korea: Statistics Korea; 2020 [cited 2021 Dec 18]. Available from: http://kostat.go.kr/portal/korea/kor_nw/1/1/index.board?bmode=read&aSeq=385322.
2. World Health Organization. Lexicon of alcohol and drug terms [Internet]. Geneva, Switzerland: World Health Organization; 1994 [cited 2021 Dec 18]. Available from: https://apps.who.int/iris/handle/10665/39461.
3. Nobili A, Pasina L, Tettamanti M, Lucca U, Riva E, Marzona I, et al. Potentially severe drug interactions in elderly outpatients: results of an observational study of an administrative prescription database. J Clin Pharm Ther 2009;34:377–86.


4. Jyrkka J, Enlund H, Lavikainen P, Sulkava R, Hartikainen S. Association of polypharmacy with nutritional status, functional ability and cognitive capacity over a three-year period in an elderly population. Pharmacoepidemiol Drug Saf 2011;20:514–22.


5. Herr M, Robine JM, Pinot J, Arvieu JJ, Ankri J. Polypharmacy and frailty: prevalence, relationship, and impact on mortality in a French sample of 2350 old people. Pharmacoepidemiol Drug Saf 2015;24:637–46.


6. Mortazavi SS, Shati M, Keshtkar A, Malakouti SK, Bazargan M, Assari S. Defining polypharmacy in the elderly: a systematic review protocol. BMJ Open 2016;6:e010989.



7. Jang IY, Lee HY; Lee E; 50th Anniversary Committee of Korean Geriatrics Society. Geriatrics Fact Sheet in Korea 2018 From National Statistics. Ann Geriatr Med Res 2019;23:50–3.



8. Nam YS, Han JS, Kim JY, Bae WK, Lee K. Prescription of potentially inappropriate medication in Korean older adults based on 2012 Beers Criteria: a cross-sectional population based study. BMC Geriatr 2016;16:118.



9. National Institutes of Health. Osteoporosis prevention, diagnosis, and therapy [Internet]. Bethesda: National Institutes of Health; c2021 [cited 2021 May 3]. Available from: https://consensus.nih.gov/2000/2000osteoporosis111html.htm.
10. Korean Society for Bone and Mineral Research. Osteoporosis and osteoporotic fracture fact sheet 2019 [Internet]. Seoul, Korea: Korean Society for Bone and Mineral Research; 2019 [cited 2021 Dec 18]. Available from: https://www.ksbmr.org/bbs/index.html?code=fact&category=&gubun=&page=1&number=703&mode=view&keyfield=&key=.
11. Korean Society of Lipid and Atherosclerosis. Dyslipidemia fact sheet in Korea 2020 [Internet]. Seoul, Korea: Korean Society of Lipid and Atherosclerosis; 2020 [cited 2021 Dec 18]. Available from: https://www.lipid.or.kr/file/Dyslipidemia_Fact_Sheet_2020.pdf.
12. Leutner M, Matzhold C, Bellach L, Deischinger C, Harreiter J, Thurner S, et al. Diagnosis of osteoporosis in statin-treated patients is dose-dependent. Ann Rheum Dis 2019;78:1706–11.


13. Hollingworth S, Duncan EL, Martin JH. Marked increase in proton pump inhibitors use in Australia. Pharmacoepidemiol Drug Saf 2010;19:1019–24.


14. Hussain S, Siddiqui AN, Habib A, Hussain MS, Najmi AK. Proton pump inhibitors’ use and risk of hip fracture: a systematic review and meta-analysis. Rheumatol Int 2018;38:1999–2014.


15. Rawla P, Sunkara T, Ofosu A, Gaduputi V. Potassium-competitive acid blockers: are they the next generation of proton pump inhibitors? World J Gastrointest Pharmacol Ther 2018;9:63–8.



16. Eguchi H, Tsuruta M, Tani J, Kuwahara R, Hiromatsu Y. Hypophosphatemic osteomalacia due to drug-induced Fanconi’s syndrome associated with adefovir dipivoxil treatment for hepatitis B. Intern Med 2014;53:233–7.


17. Bedimo R, Maalouf NM, Zhang S, Drechsler H, Tebas P. Osteoporotic fracture risk associated with cumulative exposure to tenofovir and other antiretroviral agents. AIDS 2012;26:825–31.


18. Jonklaas J, Bianco AC, Bauer AJ, Burman KD, Cappola AR, Celi FS, et al. Guidelines for the treatment of hypothyroidism: prepared by the American thyroid association task force on thyroid hormone replacement. Thyroid 2014;24:1670–751.



20. Bilezikian JP, Watts NB, Usiskin K, Polidori D, Fung A, Sullivan D, et al. Evaluation of bone mineral density and bone biomarkers in patients with type 2 diabetes treated with canagliflozin. J Clin Endocrinol Metab 2016;101:44–51.


21. National Evidence-based Healthcare Collaborating Agency. Comparative effect study on statin use and diabetes risk [Internet]. Seoul, Korea: National Evidence-based Healthcare Collaborating Agency; 2016 [cited 2021 Dec 18]. Available from: https://www.neca.re.kr/lay1/program/S1T11C145/report/view.do?seq=178.
22. Song H, Shin S, Jang S. Medical services utilization among lipid-modifying drug users. Korean J Health Promot 2011;11:25–33.
23. Choi HJ. Drug-related issues-statin (1) [Internet]. Seoul, Korea: Korea Pharmaceutical Information Center; 2014 [cited 2021 Dec 18]. Available from: http://www.health.kr/Menu.PharmReview/View.asp?PharmReview_IDX=133.
24. An T, Hao J, Sun S, Li R, Yang M, Cheng G, et al. Efficacy of statins for osteoporosis: a systematic review and meta-analysis. Osteoporos Int 2017;28:47–57.


25. Lin SM, Wang JH, Liang CC, Huang HK. Statin use is associated with decreased osteoporosis and fracture risks in stroke patients. J Clin Endocrinol Metab 2018;103:3439–48.


26. Yang YX, Lewis JD, Epstein S, Metz DC. Long-term proton pump inhibitor therapy and risk of hip fracture. JAMA 2006;296:2947–53.


27. Targownik LE, Lix LM, Leung S, Leslie WD. Proton-pump inhibitor use is not associated with osteoporosis or accelerated bone mineral density loss. Gastroenterology 2010;138:896–904.


28. Min YW, Lee YC, Kim K, Ryu S, Hong KS, Jeon HH, et al. Proton pump inhibitor use is associated with hip fracture development: a nationwide population-based cohort study. Korean J Intern Med 2020;35:1084–93.


29. Chandran M. Clinical aspects and management of osteoporosis and fragility fractures in patients with diabetes. Osteoporos Sarcopenia 2017;3:123–7.



30. Tang HL, Li DD, Zhang JJ, Hsu YH, Wang TS, Zhai SD, et al. Lack of evidence for a harmful effect of sodium-glucose co-transporter 2 (SGLT2) inhibitors on fracture risk among type 2 diabetes patients: a network and cumulative meta-analysis of randomized controlled trials. Diabetes Obes Metab 2016;18:1199–206.


- TOOLS









