2. Collard RM, Boter H, Schoevers RA, Oude Voshaar RC. Prevalence of frailty in community-dwelling older persons: a systematic review. J Am Geriatr Soc 2012;60:1487–92.


4. Kojima G. Prevalence of frailty in nursing homes: a systematic review and meta-analysis. J Am Med Dir Assoc 2015;16:940–5.


7. Figueroa JF, Joynt Maddox KE, Beaulieu N, Wild RC, Jha AK. Concentration of potentially preventable spending among high-cost medicare subpopulations: an observational study. Ann Intern Med 2017;167:706–13.


8. Joynt KE, Figueroa JF, Beaulieu N, Wild RC, Orav EJ, Jha AK. Segmenting high-cost Medicare patients into potentially actionable cohorts. Healthc (Amst) 2017;5:62–7.


10. Hoogendijk EO, Afilalo J, Ensrud KE, Kowal P, Onder G, Fried LP. Frailty: implications for clinical practice and public health. Lancet 2019;394:1365–75.


11. Dent E, Martin FC, Bergman H, Woo J, Romero-Ortuno R, Walston JD. Management of frailty: opportunities, challenges, and future directions. Lancet 2019;394:1376–86.


13. Buta BJ, Walston JD, Godino JG, Park M, Kalyani RR, Xue QL, et al. Frailty assessment instruments: systematic characterization of the uses and contexts of highly-cited instruments. Ageing Res Rev 2016;26:53–61.


21. Saliba D, Elliott M, Rubenstein LZ, Solomon DH, Young RT, Kamberg CJ, et al. The Vulnerable Elders Survey: a tool for identifying vulnerable older people in the community. J Am Geriatr Soc 2001;49:1691–9.


25. Jones DM, Song X, Rockwood K. Operationalizing a frailty index from a standardized comprehensive geriatric assessment. J Am Geriatr Soc 2004;52:1929–33.


27. Gobbens RJ, van Assen MA, Luijkx KG, Wijnen-Sponselee MT, Schols JM. The Tilburg Frailty Indicator: psychometric properties. J Am Med Dir Assoc 2010;11:344–55.


28. Nicosia FM, Spar MJ, Steinman MA, Lee SJ, Brown RT. Making function part of the conversation: clinician perspectives on measuring functional status in primary care. J Am Geriatr Soc 2019;67:493–502.


30. Lin KJ, Schneeweiss S. Considerations for the analysis of longitudinal electronic health records linked to claims data to study the effectiveness and safety of drugs. Clin Pharmacol Ther 2016;100:147–59.


36. Rockwood K, Mitnitski A. Limits to deficit accumulation in elderly people. Mech Ageing Dev 2006;127:494–6.


38. Shi SM, Kim DH. The challenges of using the Hospital Frailty Risk Score. Lancet 2019;392:2692.

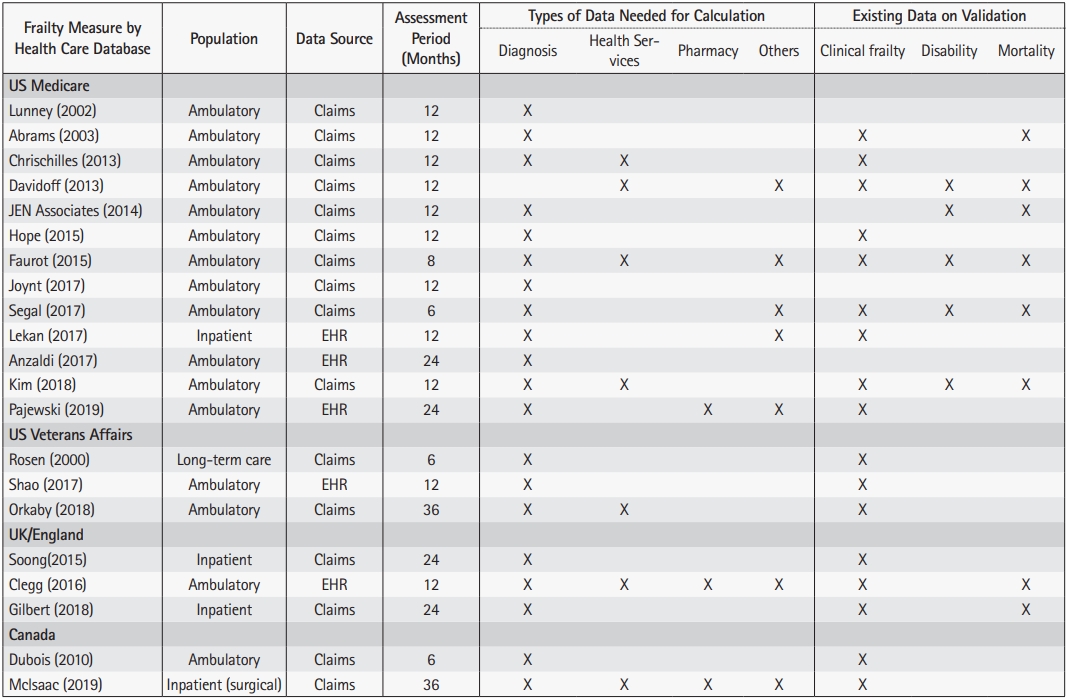
42. Lunney JR, Lynn J, Hogan C. Profiles of older medicare decedents. J Am Geriatr Soc 2002;50:1108–12.


43. Abrams C, Lieberman R, Weiner JP. Development and evaluation of the Johns Hopkins University Risk adjustment models for Medicare+Choice plan payment. Baltimore, MD: Johns Hopkins University Press; 2003.
44. Chrischilles E, Schneider K, Wilwert J, Lessman G, O'Donnell B, Gryzlak B, et al. Beyond comorbidity: expanding the definition and measurement of complexity among older adults using administrative claims data. Med Care 2014;52 Suppl 3:S75–84.


45. JEN Associates Inc. MassHealth SCO program evaluation pre-SCO enrollment period CY2004 and post-SCO enrollment period CY2005 nursing home entry rate and frailty level comparisons. Cambridge, MA: JEN Associates Inc; 2008.
46. JEN Associates Inc. JEN Frailty Index (JFI) [Internet]. Cambridge, MA: JEN Associates Inc.; c2019 [cited 2020 Feb 1]. Available from:
https://www.jen.com/jfi2.
48. Faurot KR, Jonsson Funk M, Pate V, Brookhart MA, Patrick A, Hanson LC, et al. Using claims data to predict dependency in activities of daily living as a proxy for frailty. Pharmacoepidemiol Drug Saf 2015;24:59–66.


50. Rosen A, Wu J, Chang BH, Berlowitz D, Ash A, Moskowitz M. Does diagnostic information contribute to predicting functional decline in long-term care? Med Care 2000;38:647–59.


51. McIsaac DI, Wong CA, Huang A, Moloo H, van Walraven C. Derivation and validation of a generalizable preoperative frailty index using population-based health administrative data. Ann Surg 2019;270:102–8.


52. Dubois MF, Dubuc N, Kroger E, Girard R, Hebert R. Assessing comorbidity in older adults using prescription claims data. J Pharm Health Serv Res 2010;1:157–65.

53. Soong J, Poots AJ, Scott S, Donald K, Woodcock T, Lovett D, et al. Quantifying the prevalence of frailty in English hospitals. BMJ Open 2015;5:e008456.

54. Sternberg SA, Bentur N, Abrams C, Spalter T, Karpati T, Lemberger J, et al. Identifying frail older people using predictive modeling. Am J Manag Care 2012;18:e392–7.

56. Kim DH, Patorno E, Pawar A, Lee H, Schneeweiss S, Glynn RJ. Measuring frailty in administrative claims data: comparative performance of four claims-based frailty measures in the united states medicare data. J Gerontol A Biol Sci Med Sci 2019.


61. Yao NA, Ritchie C, Cornwell T, Leff B. Use of home-based medical care and disparities. J Am Geriatr Soc 2018;66:1716–20.


65. Lekan DA, Wallace DC, McCoy TP, Hu J, Silva SG, Whitson HE. Frailty assessment in hospitalized older adults using the electronic health record. Biol Res Nurs 2017;19:213–28.


66. Lekan DA, McCoy TP. Frailty risk in hospitalised older adults with and without diabetes mellitus. J Clin Nurs 2018;27:3510–21.


69. Shao Y, Mohanty AF, Ahmed A, Weir CR, Bray BE, Shah RU, et al. Identification and use of frailty indicators from text to examine associations with clinical outcomes among patients with heart failure. AMIA Annu Symp Proc 2017;2016:1110–8.


74. Lin KJ, Singer DE, Glynn RJ, Murphy SN, Lii J, Schneeweiss S. Identifying patients with high data completeness to improve validity of comparative effectiveness research in electronic health records data. Clin Pharmacol Ther 2018;103:899–905.


78. Weiskopf NG, Rusanov A, Weng C. Sick patients have more data: the non-random completeness of electronic health records. AMIA Annu Symp Proc 2013;2013:1472–7.


79. Kharrazi H, Anzaldi LJ, Hernandez L, Davison A, Boyd CM, Leff B, et al. The value of unstructured electronic health record data in geriatric syndrome case identification. J Am Geriatr Soc 2018;66:1499–507.


80. McIsaac DI, Bryson GL, van Walraven C. Association of frailty and 1-year postoperative mortality following major elective noncardiac surgery: a population-based cohort study. JAMA Surg 2016;151:538–45.


81. McIsaac DI, Beaule PE, Bryson GL, Van Walraven C. The impact of frailty on outcomes and healthcare resource usage after total joint arthroplasty: a population-based cohort study. Bone Joint J 2016;98-B:799–805.


82. McIsaac DI, Moloo H, Bryson GL, van Walraven C. The association of frailty with outcomes and resource use after emergency general surgery: a population-based cohort study. Anesth Analg 2017;124:1653–61.


83. McIsaac DI, Wijeysundera DN, Huang A, Bryson GL, van Walraven C. Association of the hospital volume of frail surgical patients cared for with outcomes after elective, major noncardiac surgery: a retrospective population-based cohort study. Anesthesiology 2017;126:602–13.


84. Tan HJ, Shirk JD, Chamie K, Litwin MS, Hu JC. Patient function and the value of surgical care for kidney cancer. J Urol 2017;197:1200–7.


86. De Jonge KE, Jamshed N, Gilden D, Kubisiak J, Bruce SR, Taler G. Effects of home-based primary care on Medicare costs in high-risk elders. J Am Geriatr Soc 2014;62:1825–31.


87. Soong J, Poots AJ, Scott S, Donald K, Bell D. Developing and validating a risk prediction model for acute care based on frailty syndromes. BMJ Open 2015;5:e008457.

88. Hui Y, van Walraven C, McIsaac DI. How do common comorbidities modify the association of frailty with survival after elective noncardiac surgery? A Population-Based Cohort Study. Anesth Analg 2019;129:1699–706.


89. Rosen A, Wu J, Chang BH, Berlowitz D, Rakovski C, Ash A, et al. Risk adjustment for measuring health outcomes: an application in VA long-term care. Am J Med Qual 2001;16:118–27.


91. Feliciano J, Gardner L, Hendrick F, Edelman MJ, Davidoff A. Assessing functional status and the survival benefit of chemotherapy for advanced non-small cell lung cancer using administrative claims data. Lung Cancer 2015;87:59–64.