An Unusual Case of Shock in an Octogenarian
Article information
Abstract
An octogenarian presented to our hospital in shock after being “found down” at home. She was dehydrated, hypernatremic, and suffering from a urinary tract infection. Findings of a murmur and those on electrocardiography led to the performance of echocardiography, with all findings consistent with a diagnosis of hypertrophic obstructive cardiomyopathy. The patient was volume resuscitated and administered antibiotics; after stabilization, she was started on a low-dose beta-blocker. Hypertrophic cardiomyopathy is a common genetic disorder that is usually diagnosed in the second to fifth decades of life, rarely being diagnosed at an advanced age. It is also an uncommon cause or contributor to shock. We briefly review the diagnosis and management of hypertrophic cardiomyopathy in older adults, particularly in the setting of shock.
INTRODUCTION
The differential diagnosis of patients presenting with shock is broad and includes sepsis, hypovolemia, anaphylaxis, neurogenic compromise, impaired cardiac output from a low ejection fraction, and tamponade, among other causes. Although hypertrophic cardiomyopathy (HCM) is common in the general population, it is an uncommon cause of or contributor to shock. It is also usually diagnosed in younger individuals. The condition is increasingly diagnosed in older patients because of an increased index of suspicion and use of imaging. However, diagnosing an octogenarian with HCM is uncommon and may be a complex endeavor. We report a case of an initial diagnosis of HCM in a patient who presented with shock and review pertinent diagnostic, prognostic, and management considerations.
CASE REPORT
An 84-year-old woman was brought to the emergency department (ED) after being found at home (where she lived independently) by her son with whom she had not had any contact for 4 days. In the ED, she was found to have a heart rate of 103 bpm, blood pressure (BP) of 83/53 mmHg, and oxygen saturation of 96% on ambient air. She had very dry mucous membranes, and a 4/6 systolic murmur was best heard at the base and in her bilateral carotid arteries. Laboratory examinations showed a sodium level of 165 mEq/L, a creatinine level of 5.4 mg/dL, an international normalized ratio (INR) of 3.2, an elevated white blood cell count, and urinalysis findings consistent with an infection (culture grew pan-sensitive Escherichia coli). Computed tomography of the head revealed no remarkable findings. Her son reported that she had no medical or surgical history, was not taking medication, and had not seen a healthcare provider in years. He denied her having any “bad habits” or a family history of medical problems. Written informed consent was obtained from the patient for publication of this case report and accompanying images.
The patient received 2 L of lactated Ringer’s solution, and antibiotic therapy was initiated. Her BP increased to 107/64 mmHg; her mental status also improved (able to answer yes/no questions), and she was able to follow basic instructions. Because of the murmur observed and the findings on electrocardiography, she underwent echocardiography (Fig. 1). The results of electrocardiography and echocardiography were consistent with a finding of HCM. Specifically, the electrocardiogram (ECG) showed a high voltage and repolarization abnormalities consistent with left ventricular hypertrophy. The echocardiogram showed severe asymmetric septal predominant hypertrophy (septal wall thickness, 1.9 cm; posterior wall thickness, 0.97 cm), systolic anterior motion of the mitral valve with outflow tract obstruction, severe peak and mean left ventricular outflow tract gradients (248 and 130 mmHg, respectively), and moderate mitral regurgitation.
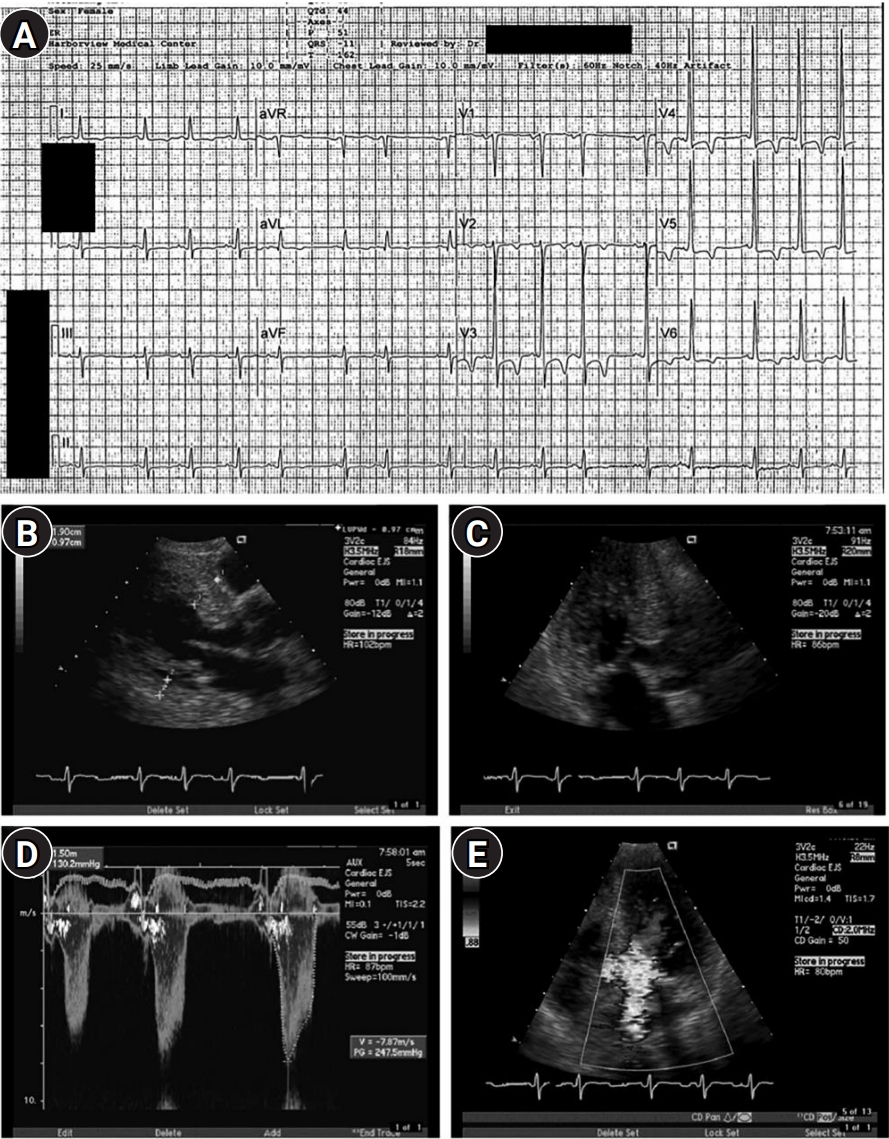
(A) Electrocardiogram obtained at admission showing normal sinus rhythm with premature atrial complexes, as well as voltages and repolarization abnormalities consistent with left ventricular hypertrophy. The remaining panels show findings consistent with hypertrophic obstructive cardiomyopathy, including (B) predominant septal hypertrophy, (C) systolic anterior motion of the mitral valve, (D) a very high left ventricular outflow tract gradient, and (E) moderate mitral regurgitation. Other findings included a normal-sized left ventricle with hyperdynamic function (ejection fraction >70%).
DISCUSSION
HCM is a common genetic condition affecting at least one in 500 persons.1) Although the condition affects people of all ages, most patients are diagnosed early in life (mean age, 44 years). HCM is increasingly diagnosed in older adults, likely because of an increasing index of suspicion as well the growing use of imaging, which may uncover the phenotype in patients being evaluated for cardiac and non-cardiac conditions (e.g., during echocardiography, chest computed tomography, or magnetic resonance imaging [MRI]). When diagnosed in older adults, the condition may be late-onset or, because of a lack of symptoms, late to have been recognized.
When an older adult presents with symptoms that may be attributable to HCM, the differential diagnosis is extensive. Dyspnea on exertion, chest pain, and syncope can have many etiologies or be multifactorial owing to age-related problems, including volume depletion, deconditioning, or comorbidities such as lung conditions (e.g., chronic obstructive pulmonary disease), coronary artery disease, valve disease, arrhythmias, or heart failure. Physical examination may be challenging, as the murmur due to HCM may be confused with or masked by that due to aortic sclerosis or stenosis. These other etiologies may be pursued before cardiac imaging (usually echocardiography). Once imaging raises the possibility of a differential diagnosis of HCM, the diagnosis may remain uncertain as other conditions can have similar morphologic appearances.
Other entities that may be considered include age-related structural changes (discussed below), hypertensive cardiomyopathy, and amyloid or other infiltrative cardiomyopathies (e.g., hemochromatosis). A detailed approach to the diagnosis of these is beyond the scope of this report; however, more global hypertrophy and a history of un/under-treated hypertension and other end-organ damage suggest hypertensive cardiomyopathy and hypertrophy on echocardiography but a low voltage on ECG, whereas other specific findings on imaging or comorbid conditions may point toward amyloid or other infiltrative cardiomyopathy.
A sigmoid-shaped septum (also called discrete upper septal thickening or upper septal knuckle) is found in older adults (particularly those with hypertension) and may result from normal aging. The prevalence is up to 10%, and an increased angle of the aorta to the long axis of the left ventricle (LV) is also common. In the Framingham Heart Study, 1.5% of 3,562 participants had a sigmoid-shaped septum (17.8% of those >85 years). The positive correlates were increasing age, hypertension, decreased LV fraction, and mitral annular calcification, whereas the inverse correlates were the LV diastolic dimension. After adjusting for cardiovascular risk factors, over 15 years, the condition was not found to be associated with cardiovascular events or mortality.2)
Differentiating normal aging from genetic HCM is challenging. Patients with late-onset HCM tend to have less impressive septal hypertrophy and an ovoid or ellipsoid (not crescentic) LV.3) Other differences include narrower left ventricular outflow tracts (LVOTs), anterior displacement of the mitral valve, and a larger area of septum-anterior leaflet contact.4) Chen-Tourneaux et al.5) suggested that specific echo-Doppler findings distinguish the sigmoid septum from HCM with a sensitivity of 78% and a specificity of 90%; moreover, cardiac MRI is also increasingly used for evaluation.
Of the 3% of patients in the Framingham Heart Study (n=1,862; mean age, 60 years) with unexplained LV hypertrophy, only 18% had sarcomeric contractile protein mutations.6) Another analysis reported that the sites of mutation found in late-onset patients (myosin binding protein-C, troponin I, alpha cardiac myosin heavy chain) differed from those found in younger patients (beta-myosin heavy chain, troponin T, alpha tropomyosin).7)
The clinical course of patients diagnosed with HCM at an advanced age is generally better than that of those diagnosed at a younger age.8,9) In a study of 428 patients aged ≥60 years followed up for nearly 5 years, the rates of HCM-related morbidity and mortality (including sudden death) were low. Other studies have also shown lower rates of traditional HCM-related risk factors for sudden cardiac death (SCD)10,11) but concomitantly higher rates of atherosclerotic cardiovascular risk factors in older cohorts.12) Of the 428 patients included in that study, only 16 had HCM-related mortality (0.64% per year).13) Death from progressive heart failure occurred in only two patients, embolic stroke in six patients, and SCD in five (1.2% of the population; 0.20% per year). Other reports also reveal that the HCM-related rates of heart failure, embolic stroke, or SCD in patients >60 years of age are lower than those reported at younger ages, with annual mortality rates less than those in age- and sex-matched controls.13,14) Notably, the presence of atherosclerotic risk factors, atherosclerosis itself, and other comorbidities in this population contributed to the finding that older HCM patients most frequently die of non-HCM related causes.13)
Standard treatment may be considered for older adults diagnosed with HCM, including medical therapy (e.g., beta-blockers, calcium channel blockers, and disopyramide). Medically refractory patients, even those of advanced age, may be evaluated for alcohol septal ablation or surgical septal myectomy.12) Family/genetic counseling and testing should be considered, as well as evaluation for primary or secondary prevention ICD (implantable cardioverter-defibrillator) placement and a rhythm control strategy for patients with atrial fibrillation that can affect up to one-third of older HCM patients (antiarrhythmic medications and/or ablation) may be contemplated, although older adults may be at an increased risk for complications.12,15) Regarding the management of patients with hypertrophic cardiomyopathy and shock, other etiologies of shock should be evaluated and treated, including infection (sepsis), anemia, and volume depletion. Echocardiography is the fastest route to the diagnosis of HCM and can be useful for evaluating and managing other etiologies of shock; thus, this should be considered early after presentation. Lastly, to avoid worsening LVOT obstruction, both volume depletion and inotropes should be avoided. If vasopressors are required (they were not in the present case), choosing one that is not also an inotrope is advisable.
In conclusion, after correction of the free water deficit and the administration of antibiotics, the patient’s laboratory abnormalities resolved and her mental status returned to baseline. Her shock was likely multifactorial, owing to a combination of dehydration and urinary tract infection, resulting in volume depletion and increased myocardial demand, both of which worsened the LVOT obstruction, further impairing her cardiac output. After stabilization, the patient was started on a low-dose beta-blocker in addition to antibiotics. She and her son declined further evaluation of her HCM (including cardiac MRI, genetic evaluation, and outpatient rhythm monitoring for ventricular arrhythmias/risk stratification for sudden death), and she was discharged to a rehabilitation facility with plans for outpatient follow-up.
Notes
CONFLICT OF INTEREST
The researcher claims no conflicts of interest.
FUNDING
None.
