Relationships between Spinal Sarcopenia and Spinal Sagittal Balance in Older Women
Article information
Abstract
Background
Spinal sarcopenia is receiving renewed attention as a cause of spinal sagittal imbalance. However, the relationships between spinal sarcopenia and spinal sagittal balance (SSB) have not been thoroughly investigated. We evaluated the relationships between SSB parameters and sarcopenic indices with lumbar paraspinal muscle (LPM) quantity and strength in healthy older adults.
Methods
Twenty-four healthy community-dwelling older women were enrolled. Demographic variables, conventional sarcopenic indices, isometric back muscle strength, and SSB parameters, as well as results of functional examinations and lumbar spine computed tomography scan with LPM cross-sectional area (CSA) and density assessments, were examined. The independent effect on the sum of the total LPM CSA was determined using multivariable regression analysis adjusted for age, appendicular skeletal muscle mass, gait speed, handgrip strength, back extensor strength, and pelvic tilt (PT) angle.
Results
PT angle was significantly correlated with the sum of the total LPM CSA and mean LPM density (r=-0.502, p=0.015 and r=0.504, p=0.014, respectively). Furthermore, PT angle was an independent factor for the sum of the total LPM CSA (β=-0.610, p=0.021) in the multivariate regression models (R2=0.320).
Conclusion
Our data suggest that PT angle was significantly correlated with LPM CSA in healthy older women. To our knowledge, this is the first report to investigate the relationships of sarcopenic indices and spinal muscle degeneration with SSB.
INTRODUCTION
The spine is an inevitable site of sarcopenia owing to the large muscles surrounding it. Sarcopenia of the lumbar paraspinal muscles (LPMs) has been receiving renewed attention as a cause of spinal degeneration. Both atrophy and fatty changes in paraspinal muscles originating from sarcopenia are associated with functional disorders and chronic back pain.1)
Conventional indices to define sarcopenia—appendicular skeletal muscle mass (ASM), handgrip strength (HGS), and gait speed—cannot reflect regional sarcopenia and its clinical outcomes. A cross-sectional study of 821 subjects with knee osteoarthritis and 4,103 controls showed that low skeletal muscle mass in the lower limbs was correlated with the presence of knee osteoarthritis, whereas whole-body skeletal muscle mass was not.2) The authors suggested the need for limb-specific muscle mass examinations to assess the effects of skeletal muscles on a specific joint. Therefore, regional measurements should be performed to evaluate the outcome of sarcopenia in focal areas.
However, there are few simple and clinically valid measuring tools to assess sarcopenia in the spine. Whole-body dual-energy X-ray absorptiometry and bioimpedance analysis (BIA) to measure ASM cannot be applied to spinal sarcopenia; thus, tomographic imaging such as computed tomography (CT) and magnetic resonance imaging (MRI) are required to measure paraspinal muscle quantity. Moreover, tools to verify the function and performance of paraspinal muscle are more difficult to evaluate. To assess paraspinal muscle strength, expensive special equipment such as isokinetic dynamometry is required. Furthermore, there is no standardized test for spinal muscle performance.
Spinal sagittal balance (SSB) is an important indicator of outcomes of lumbar spine surgery3) and non-operative treatment of spinal stenosis.4) While SSB can be affected by sex5) and ethnicity,6) aging itself is the most important cause of spinal sagittal imbalance.7) LPM degeneration is one of the causes of spinal sagittal imbalance. An MRI-based study reported the relationship between the estimated LPM volume and sagittal curvature magnitude.8) One cross-sectional study suggested that the muscle thickness of the erector spinae and echo intensity of the lumbar multifidus were independent variables of SSB.1)
However, the relationships between spinal sarcopenia and SSB have not been thoroughly investigated. Thus, we evaluated the relationships of SSB parameters and sarcopenic indices with LPM quantity and strength in healthy older adults. We hypothesized that SSB parameters could reflect LPM mass and back extensor strength.
MATERIALS AND METHODS
Study Population
Healthy community-dwelling older (≥65 years) women who could independently walk more than 100 m were consecutively enrolled in a single center from July 2018 to December 2018. Participants who had experienced the following were excluded: (1) low back pain with moderate severity (numeric rating scale 5 and over); (2) history of any type of lumbar spine surgery; (3) history of hip fracture surgery and arthroplasty of the hip or knee; (4) disorders of the central nervous system such as stroke, parkinsonism, or spinal cord injury; (5) communication disorder such as severe hearing loss; (6) musculoskeletal conditions affecting physical function such as limb amputation; (7) long-term use of corticosteroids due to inflammatory disease; (8) malignancy requiring treatment within 5 years; and (9) other medical conditions requiring active treatment; additionally, individuals who refused to participate in the study were also excluded.
Conventional Sarcopenia Work-Up
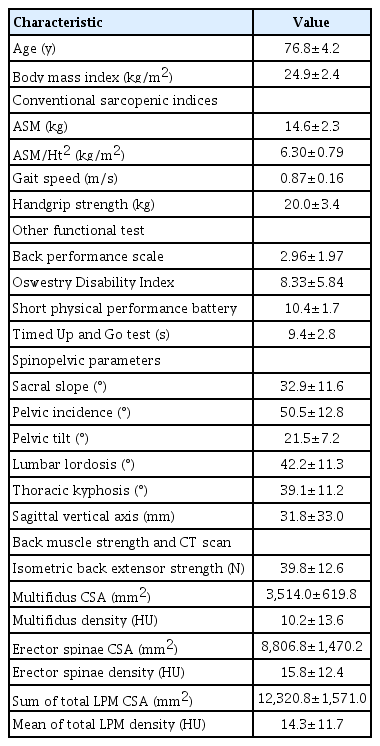
BIA (InBody 720; InBody, Seoul, Korea) was used to analyze body composition including lean body and fat masses. ASM was calculated as the sum of the lean mass in the bilateral upper and lower extremities9) and standardized by dividing by the squared height (ASM/Ht2, kg/m2). HGS was measured using a hand-grip dynamometer (T.K.K.5401; Takei Scientific Instruments, Tokyo, Japan),10) as described previously.11) Briefly, participants were asked to perform the following while sitting in a straight-backed chair with their feet flat on the floor: adduct and neutrally rotate the shoulder, flex the elbow to 90°, and place the forearm in a neutral position with the wrist between 0° and 30° extension and between 0° and 15° ulnar deviation. Participants were instructed to squeeze the handle as hard as possible for 3 seconds, and the maximum contraction force (kg) was recorded. Gait speed was measured using a 6-meter usual gait speed (m/s) as recommended by the Asian Working Group for Sarcopenia. The definition and cutoffs for sarcopenia were also adopted from these guidelines.12)
Functional Examinations and Questionnaires
Functional examination using a short physical performance battery (SPPB) was derived from three objective physical function tests (i.e., time taken to cover 4 m at a comfortable walking speed, time taken to stand from sitting in a chair five times without stopping, and ability to maintain balance for 10 seconds in three different foot positions at progressively more challenging levels).13) A score from 0 to 4 was assigned to the performance of each task, with higher scores indicating better lower body function. The Timed Up and Go test (TUG) has shown excellent test–retest reliability in older adults.14) The participants were provided with verbal instructions to stand up from an armchair, walk 3 m as fast as possible, turn back at a cone set out by the researchers, walk back, and sit down in the chair. They were allowed to wear their regular footwear and use a walking aid if needed. A stopwatch was started on the word ‘go’ and stopped when the participant was completely seated with their back against the backrest. The time to complete the test was recorded in three consecutive trials using the first trial to familiarize the participants with the test. The best time from the three trials was analyzed.15) The Oswestry Disability Index (ODI) is one of the most commonly used instruments for measuring disability in spinal disorders. It consists of 10 items that assess the level of pain and interference with several physical activities. We used the Korean version of the ODI.16) The Back Performance Scale (BPS) consists of five tests: sock test, pick-up test, roll-up test, fingertip-to-floor test, and lift test. The five tests comprising the BPS demonstrate associations with each other and each test contributes to the high internal consistency, implying that the tests share a common characteristic in measuring physical performance.17) The BPS sum score (0–15) is calculated by adding the individual scores for the five tests.
Isometric Back Muscle Strength
We measured the isometric back extensor strength using a hand-held dynamometer (PowerTrack II; JTECH Medical, Salt Lake City, UT, USA).18) Briefly, the participants stood in full extension with their backs to a wall and feet flat on the floor with heels touching the wall. An inelastic belt was looped through the anchor rails and secured firmly 1 cm below the anterior superior iliac spine to restrain movement and maintain participant contact with the wall during the test. The participants were instructed to flex forward approximately 15° at the hips so that the dynamometer could be positioned posterior to the spinous process of the 7th thoracic vertebrae. In this way, counter pressure was provided by the fixed wall behind the participants’ back to avoid tester-induced variations in resistance. Although this method is novel, it showed a strong positive relationship with back extensor strength measured using the gold-standard isokinetic dynamometry and high inter-instrument validity and reliability.19)
Spinal Sagittal Balance
For each patient, one lateral radiograph of the whole spine was obtained and digitized. All measurements were performed using imaging software (INFINITT PACS M6; INFINITT Healthcare, Seoul, Korea), as previously described.20,21) The following spinopelvic radiographic parameters were analyzed: sacral slope (SS), pelvic incidence (PI), pelvic tilt (PT), lumbar lordosis (LL), thoracic kyphosis (TK), and sagittal vertical axis (SVA).
LPM Measurement by Spine CT Scan
Lumbar spine CT scans (Ingenuity CT; Philips Healthcare, Cleveland, OH, USA) were performed to measure the cross-sectional area (CSA) and mean density (in Hounsfield unit [HU]) of the LPM (multifidus [MF] and erector spinae [ES]).22) The mean density reflected the degree of intramuscular fat content because the values decreased as the fat content increased. Before each CT scan, a calibration was performed using air as the standard. CT scanning was performed with the participant in the supine position with a 120-kV and 140-mA protocol. Using 1-mm thin-section axial CT scan images, the axial images were reformatted with each lumbar intervertebral disc level (T12/L1-L5/S1) parallel to the adjacent vertebral endplates. These axial images at each intervertebral disc level were reconstructed at 2.5-mm intervals, which included cross-sectional images of LPM. The measurement of ES and MF was performed from the level of L1/L2 to L5/S1 using a specially designed radiological workstation (MEDIP; MEDICALIP, Seoul, Korea) (Fig. 1). The CSA was measured by manually constructing points around the outer margins of the individual muscles using a touchscreen LCD monitor (XPS 15 9570; Dell Inc., Round Rock, TX, USA) and digital touchscreen pen (PN556W Dell Active Pen; Dell Inc.). After the CSA and mean density of paraspinal muscles were separately measured on the bilateral sides, the mean and sum values at all levels were calculated.23)
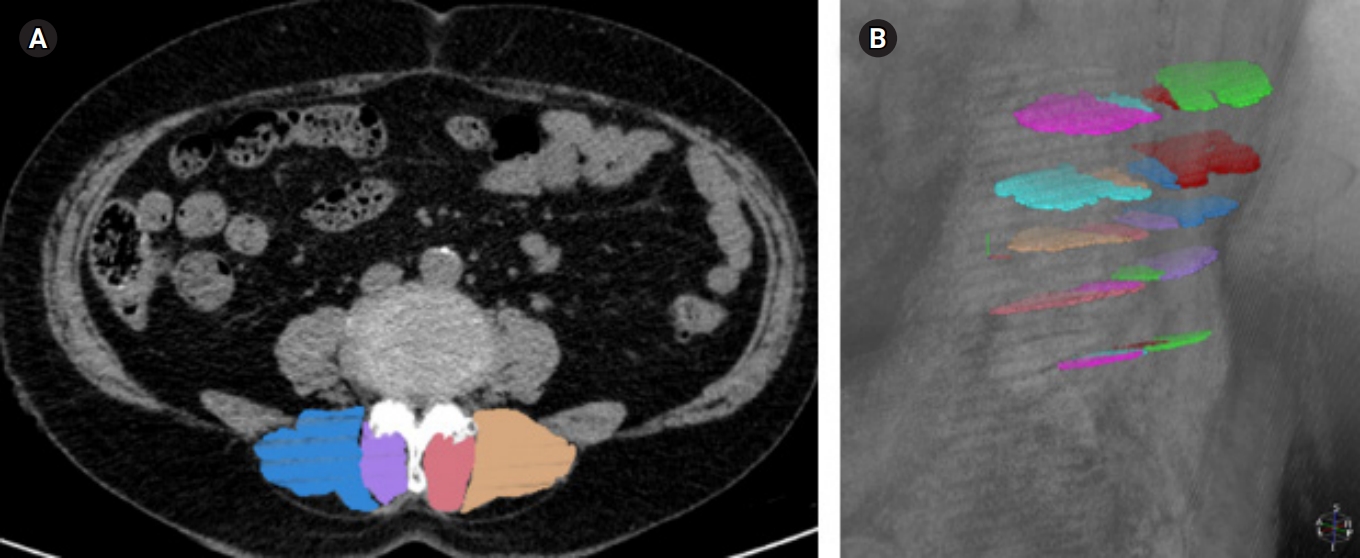
Measurements of cross-sectional area and density of lumbar paraspinal muscle. (A) Computed tomography axial image at the L3/4 disc level and (B) three-dimensional reconstructed bird’s-eye view from the top-left of the participant.
All demographic and clinical data, including CT scan images, were obtained with the approval of the Institutional Review Board of SMG-SNU Boramae Medical Center (No. 16-2017-45). Written informed consent was obtained from all participants.
Statistical Analysis
The relationships between sarcopenic indices and functional outcomes with SSB parameters were measured by Pearson correlation coefficients. The independent effects on the sum of the total LPM CSA were determined using multivariate regression models adjusted for six key factors: age,7) ASM/Ht2, HGS, gait speed,1) back extensor strength,24) and PT angle. ASM/Ht2 and HGS were included because they are the basic variables for diagnosing sarcopenia and PT was included in the regression analysis as the most relevant variable of SSB. An adjusted model was developed through backward elimination with a significance level of 0.2 to enter and 0.05 to retain. We also evaluated possible multiple collinearities between covariates by correlation analysis and collinearity statistical tests (tolerance and variance inflation factor tests) during regression analysis. IBM SPSS Statistics version 21.0 for Windows (IBM Corp., Armonk, NY, USA) was used for all analyses. p-values of less than 0.05 were considered statistically significant.
RESULTS
The baseline characteristics of 24 older women are shown in Table 1. Their mean age was 76.8±4.2 years. Among conventional sarcopenic indices, the ALM/Ht2, HGS, and gait speed were 6.30±0.79 kg/m2, 20.0±3.4 kg, and 0.87±0.16 m/s, respectively. No participant met the diagnostic criteria for sarcopenia.
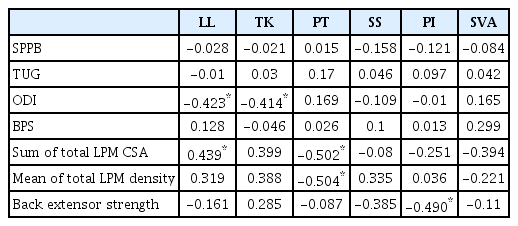
Among the conventional sarcopenic indices, only HGS was positively correlated with SPPB (r=0.521, p=0.011). While both HGS and ASM/Ht2 tended to have positive correlations with LPM mean density and back extensor strength, they were not significant (Table 2). Among SSB parameters, PT angle was significantly correlated with the sum of the total LPM CSA and mean LPM density (r=-0.502, p=0.015 and r=0.504, p=0.014, respectively) (Fig. 2). The LL angle was also correlated with the ODI and the sum of the total LPM CSA (r=-0.423, p=0.045 and r=0.439, p=0.036, respectively). Only PI angle was significantly correlated with back extensor strength (r=-0.490, p=0.018) (Table 3).

Scatter grams showing the relationships between pelvic tilt angle and LPM CSA (A) and mean density (B). LPM, lumbar paraspinal muscle; CSA, cross-sectional area.
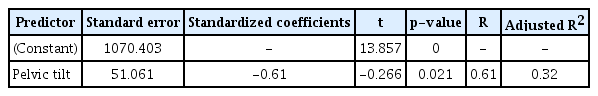
Finally, among the six key variables, multivariate regression models adjusted by the other variables revealed PT angle to be an independent factor for the sum of the total LPM CSA (β=-0.610, p=0.021, R2=0.320) (Table 4).
DISCUSSION
The most important finding of this study was that only PT angle was significantly correlated with the sum of the total LPM CSA in healthy older women. Because we used multivariate regression models adjusted for potential factors, including age and ASM, SSB parameters might be independent factors affecting spinal sarcopenia.
In the current study, conventional sarcopenic indices (ASM, HGS, and gait speed) were not correlated with LPM mass, back extensor strength, and spine specific functional outcomes. Even ASM was not correlated with LPM CSA (r=0.181, p=0.535). Therefore, conventional muscle mass measurements that sum limb muscle masses to define sarcopenia do not reflect the clinical features of spinal sarcopenia. Jeon et al.25) reported that low limb muscle mass was correlated with only knee joint radiological degeneration and not hip or spine. Therefore, site-specific muscle mass investigation is necessary to evaluate the effect of skeletal muscle on specific regions.
Among the several SSB parameters, only PT angle was significantly correlated with both LPM quantity (CSA) and quality (density), while LL angle was only correlated with LPM quantity. PT is the angle between a vertical line originating at the center of the femoral head and a line starting from the center of the femoral head to the midpoint of the endplate of S1. In simple terms, this angle describes the rotation of the pelvis around the femoral heads. PT increases with age, and high PT is needed to maintain an upright posture to compensate for kyphosis.26) While other SSB parameters such as LL and SVA can be easily affected by posture and position, The PT is a reproducible and reliable measure of global sagittal alignment regardless of the level of training.27,28) PT is also correlated with health-related quality of life in adult spinal deformity.29) Therefore, among the SSB parameters, the association of PT with spinal sarcopenia warrants further investigation.
Isokinetic back muscle strength positively affects SSB.24) A cohort study in older adults also reported the negative correlation between spinal inclination and back muscle strength (r=-0.294).30) However, in our study, back extensor strength was not independently associated with SSB, contrary to the hypothesis, although there was a simple correlation between back extensor and PI (r=-0.490). There are two potential explanation for this conflict. If the values of back extensor strengths measured in this study were normal because participants were healthy community-dwelling older women without sarcopenia, they might not affect SSB due to the ceiling effect. Another assumption was that the muscle strength measured in this study was the isometric back extensor strength, which might not be valid in older adults. Therefore, back extensor strength might be better to be evaluated using the gold-standard isokinetic dynamometer.
Our study had several limitations. First, this was a cross-sectional study and not a prospective investigation. In addition, the sample size (n=24) was not sufficient for a good prediction level in the regression model.31) Thus, the longitudinal SSB effects on spinal sarcopenia and causal relationship between SSB and spinal sarcopenia could not be verified. However, we will prospectively follow-up and evaluate the participants to answer these questions in a future study. Second, bias was possible in the participant selection. Because we enrolled only healthy and community-dwelling older women, there were no women with sarcopenia. Therefore, even though we compared sarcopenic indices to functional outcomes and SSB parameters, these outcome variables might be skewed to a healthy population and might not reflect sarcopenia and paraspinal muscle degeneration. Recently, Ohyama et al.32) reported the relationship between sarcopenia and spinopelvic parameters in 126 participants, 21.4% of whom were patients with sarcopenia. The authors reported larger SVA and TK in the sarcopenia group than those in the group without sarcopenia among patients with a spinopelvic mismatch. Thus, participants diagnosed with sarcopenia should be sufficiently included in the target population. Finally, we did not investigate the global alignment and proportion (GAP) score, which can denote ‘normal’ and ‘pathologic’ standing sagittal alignment and shape as a single score for every magnitude of pelvic incidence.33) In the GAP, the optimal sagittal alignment is based on four factors deviating from their ideal curves and these factors are proportionally related to the PI.34) Therefore, future studies should describe the SSB by measuring a single variable, such as the GAP score, rather than listing several different variables.
In conclusion, our data suggest PT angle was significantly correlated with LPM CSA in healthy older women. To our knowledge, this is the first report to investigate the relationships between sarcopenic indices and spinal muscle degeneration with SSB.
Notes
The researchers claim no conflicts of interest.
Acknowledgements
This research was funded by the Korean Geriatrics Society.