Development of a Consensus List of Potentially Inappropriate Medications for Korean Older Adults
Article information
Abstract
Background
Korea has recently attained the aged society status and the growth rate of the aging population will be among the most rapid worldwide. The objective of this study was to develop a credible list of potentially inappropriate medications (PIMs) for Korean older adults.
Methods
A new Korean PIMs list was produced through a comprehensive structured expert survey (modified Delphi method). To generate an expert panel, we invited the nomination of experts in geriatric medication from the Korean Geriatric Society, the Korean Academy of Clinical Geriatrics, the Korean Academy of Family Medicine, the Korean Association for Geriatric Psychiatry, and the Korean Association of Geriatric Hospitals. Based on their recommendation, the expert panel consisted of 14 geriatric specialists, including 10 geriatricians (7 family medicine doctors and 3 internal medicine doctors), 3 geriatric psychiatrists, and 1 clinical pharmacist. After 4 rounds, the new Korean PIMs list was finalized.
Results
Sixty-two drugs were classified as PIMs for older adults irrespective of comorbidities, including antipsychotics, tricyclic antidepressants, benzodiazepines, non-steroidal anti-inflammatory drugs, and first-generation antihistamines. Forty-eight drugs or drug categories were classified as PIMs for 18 specific conditions that older adults encounter frequently. The expert panel presented the rationale and comments including preferred therapeutic alternatives and exceptional situations for each item.
Conclusion
We presented a “user-friendly” PIMs list for Korean older adults. Further prospective studies to validate its usefulness in clinical settings and regular updating of the list are required. It is also important to disseminate this list to doctors who prescribe medication to older people.
INTRODUCTION
In January 2018, approximately 7.4 million Koreans (14.3% of the total population) were ≥65 years of age.1) Korea has just entered the aged society phase and the growth rate of this aging population is among the most rapid worldwide. The proportions of older adults ≥65 years of age are expected to be 20% in 2026, 30% in 2034, and over 40% in 2058.2)
Older patients generally use more drugs than younger people. Changes in pharmacokinetics and pharmacodynamics, poor homeostatic reserves, and increased frailty significantly increase the risk of adverse drug events (ADEs) in older populations.3) Although statistical data about ADEs in older Koreans are limited, the United Kingdom and the United States studies showed that ADEs are a common cause of morbidity and mortality in older patients. In two UK hospitals, ADEs accounted for 6.5% of hospital admissions and patients admitted with ADEs were significantly older than patients without ADEs.4) A US study also reported that ADEs are responsible for about 100,000 hospitalizations per year in adults ≥65 years of age and nearly half of these hospitalizations were among adults ≥80 years.5) In addition, the prediction and detection of ADEs is challenging in older adults and far more difficult in patients with cognitive impairment.6) Therefore the impact of ADEs could be underestimated in older populations. Furthermore, fragmented medical services in the Korean health system7) suggest that ADEs among older Koreans may be an even more serious problem compared to other countries.
Medications whose risk of ADEs outweighs their expected clinical benefit when they are administered to older persons are considered potentially inappropriate medications (PIMs), especially when there are better-tolerated alternatives.8) However, there is no widely-used, reliable PIMs list in Korea. Explicit criteria for screening PIMs that have been developed in other countries, such as the Beers criteria, are difficult to apply in Korean older people because of the differences in drug availability and prescribing behavior. The domestic PIMs list that Kim et al.9) proposed in 2010 has not been used widely because the list was developed without the collaboration of geriatric academic societies and has not been disseminated effectively to doctors who prescribe medication to older people. The objective of this study was to develop a concise PIMs list to which Korean doctors who treat older people can easily refer in their daily practice.
MATERIALS AND METHODS
A research steering team was formed to recruit experts and prepare and circulate the content of the Delphi rounds. The team comprised 2 geriatricians, a family physician, a Delphi method expert, and a public health researcher. The final PIMs list was produced through 4 rounds of comprehensive structured expert surveys distributed through email (modified Delphi method).
Expert Panel Recruitment
To generate an expert panel, we invited nomination of experts in geriatric medication from the Korean Geriatric Society, the Korean Academy of Clinical Geriatrics, the Korean Academy of Family Medicine, the Korean Association for Geriatric Psychiatry and the Korean Association of Geriatric Hospitals. All nominees were provided information regarding the study objectives and design and the commitment required for participation. Finally, the expert panel consisted of 14 geriatric specialists, including 10 geriatricians (7 family medicine doctors and 3 internal medicine doctors), 3 geriatric psychiatrists, and 1 clinical pharmacist.
First-Round Survey
The questionnaire for the first-round survey consisted of open-ended questions about the recommendations of preexisting PIMs screening tools published in English or Korean that the new Korean PIMs list should refer to, the appropriate number of items on the new list, and general opinions and comments regarding the research process.
Establishment of a Preliminary List and Second-Round Survey
According to the response of the first-round survey and literature review, a preliminary list was produced based on the 2015 Beers criteria,10) the screening tool of older person’s prescription (STOPP),11) the PRISCUS (Latin for “old and venerable”) list,12) the PIMs list for the Korean elderly by Kim et al.,9) the PIMs list from the Korean Health Insurance Review & Assessment Service,13) and the PIMs list of the Seoul National University Bundang Hospital (not published in a peer-reviewed journal). Some screening tools published before 2010 were excluded because they included duplicated or out-of-date items. The research steering team reviewed the above screening tools and matched each item with the reimbursable drugs in Korea using the KIMs online medication database. Drugs that are not available in Korea were excluded from the preliminary list. The preliminary list included 98 medications that should be avoided by most older adults and 108 medications to be avoided by older adults with 24 specific conditions.
In the questionnaire for the second-round survey, the research steering team provided summarized information from the responses to the first round and the process of developing the preliminary list. The expert panel was asked to score with 5-point Likert scales the frequency (1, very rare; 2, rare; 3, average; 4, common; 5, very common) and severity (1, trivial; 2, mild; 3, average; 4, severe; 5, very severe) of the ADEs that are potentially caused by each item in the preliminary list. The panelists were allowed a “cannot judge” option and asked to state the reason why each item was inappropriate, preferred therapeutic alternatives, methods to minimize the risk, and other general comments. To help the expert panel evaluate the items, the reference and major rationale of each item and representative brand names of the drugs were included in the questionnaire.
Establishment of the First Draft of a Korean PIMs List and Third-Round Survey
The clinical significance score (CSS) of the medications was defined as the product of the frequency and severity scores. According to the expert panel’s suggestion that the appropriate number of PIMs items would be <90, items with a CSS <9 were excluded from the first draft of the Korean PIMs list. The draft included 49 medications that should be avoided by most older adults, regardless of co-morbid conditions, and 45 medications to be avoided by older adults with specific conditions. Based on the experts’ opinions and literature review, the research steering team described the rationale and comments for each medicine on the PIMs list, including preferred therapeutic alternatives and exceptional situations. In the third-round survey, the expert panel was asked to state whether they agreed with the inclusion or exclusion of each item and if not, to present their rationale and supporting evidence.
Establishment of the Second Draft of the Korean PIMs List and Fourth-Round Survey
Reflecting the experts’ opinions in the third-round survey, the second draft of Korean PIMs list was produced. The second draft, including changes and their rationale, was submitted to the expert panel for approval. The final version of the Korean PIMs list was generated, with minor modifications of the second draft, to reflect the response to the fourth-round survey. Items for which no consensus was achieved in the fourth round were excluded from the final version.
Ethics Statement
The present study protocol was reviewed and approved by the Research Committee of Seoul Medical Center and the study was conducted in accordance with the principles of the Declaration of Helsinki.
RESULTS
The final PIMs list is presented in Tables 1 and 2. The list targets older adults outside palliative care and includes medications that most older adults (Table 1) and older adults with specific conditions (Table 2) should avoid. The expert panel emphasizes that the medications in this list are not “absolutely contraindicated” but “potentially inappropriate”; thus, the medications can be used cautiously for older adults if clinicians judge that the potential benefit outweighs the risk.
PIMs for Older Adults (Table 1)
A total of 62 drugs were classified as PIMs for older adults irrespective of comorbidities, most of which were antipsychotics, tricyclic antidepressants (TCA), benzodiazepines, first-generation antihistamines, and nonsteroidal anti-inflammatory drugs (NSAID). Some antiparkinsonian drugs (benztropine, trihexyphenidyl), antispasmodics (clidinium-chlordiazepoxide, scopolamine), skeletal muscle relaxants (methocarbamol, orphenadrine), and oxybutynin were included in this list due to their strong anticholinergic properties. Among cardiovascular drugs, some antiarrhythmics (dronedarone, amiodarone, and flecainide), digoxin, and ticlopidine were included due to their worse clinical outcomes or higher risk of adverse events compared to those of available alternatives.
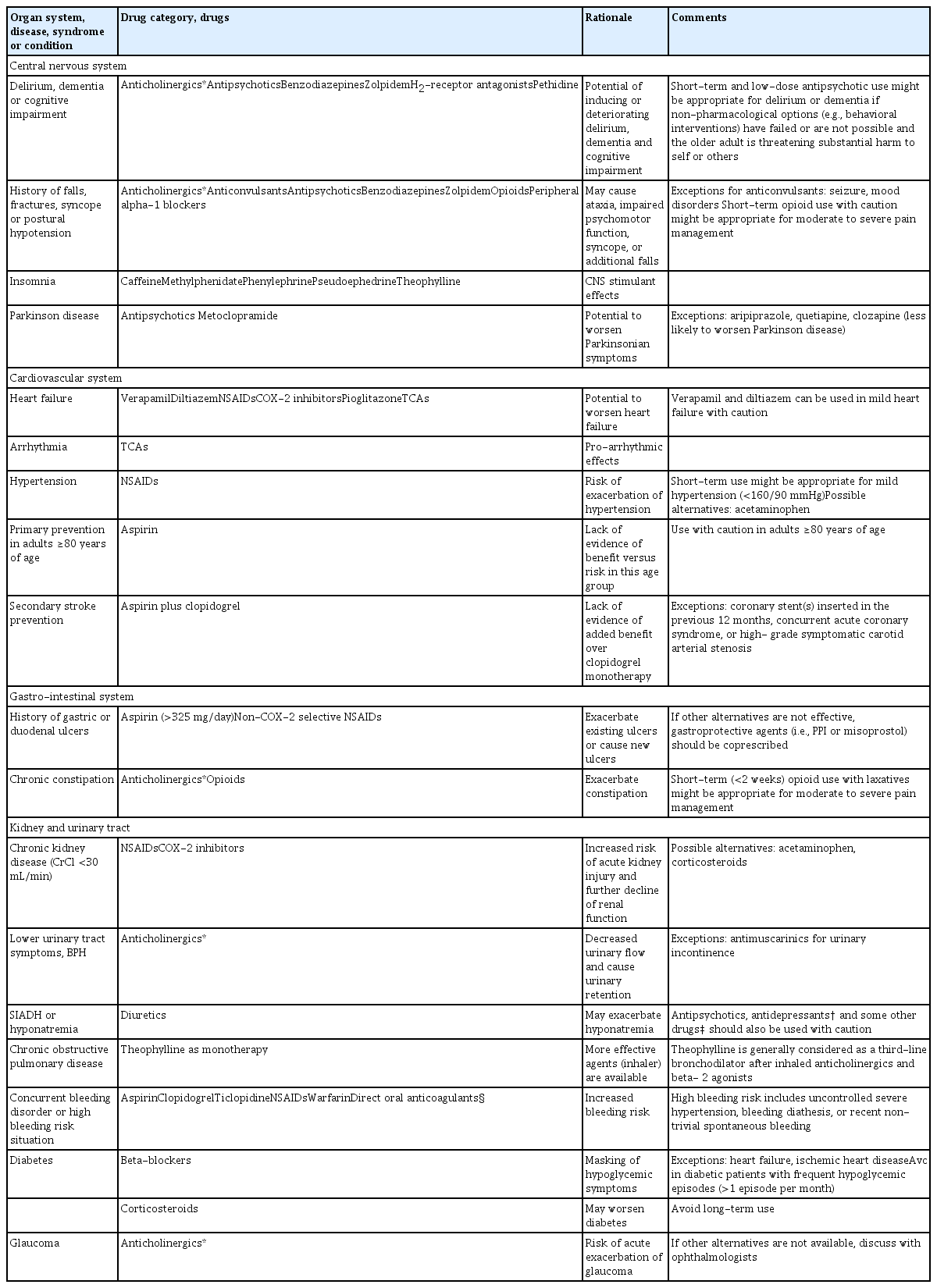
PIMs in Older Adults With Specific Conditions (Table 2)
A total of 48 drugs or drug categories were classified as PIMs for 18 specific conditions that older adults encounter frequently. The panel agreed that anticholinergics are especially inappropriate for older adults with cognitive problems, higher fall risk, constipation, lower urinary tract symptoms, or glaucoma, which includes first-generation antihistamines, bladder antimuscarinics, tricyclic antidepressants, and some antipsychotics. Benzodiazepine and zolpidem were also classified as PIMs for older patients with cognitive problems and higher fall risk. Antipsychotics and metoclopramide should be avoided in patients with Parkinson disease whenever possible. The panel agreed that NSAID use requires special precautions in older adults with heart failure, hypertension, chronic kidney disease, history of peptic ulcer, or concurrent bleeding disorders. The use of aspirin in older adults aged ≥80 years for cardiovascular primary prevention and the use of aspirin plus clopidogrel for stroke secondary prevention are discouraged except in special situations because they could increase bleeding risk without definite benefit. Hyponatremic older patients are recommended to avoid diuretics, some antidepressants and antipsychotics.
DISCUSSION
We present a new PIMs list for Korean older adults that was developed in collaboration with major geriatric academic societies in Korea. The list was generated with the modified Delphi method referring to recently published international screening tools such as the 2015 Beers criteria and the revised STOPP.
A prospective, observational cohort study showed that the self-reported history of falls (odds ratio, 1.23; 95% confidence interval [CI], 1.04–1.45) and hospital admission (hazard ratio, 1.16; 95% CI, 1.08–1.24) were independently associated with the use of PIMs in community-dwelling older men.14) Hamilton et al.15) also showed in a European prospective study that the prescription of PIMs according to the original STOPP criteria was significantly associated with ADEs that cause or contribute to hospital admission in older adults. With the rapid increase of older populations, several studies regarding PIMs have been also published in Korea. Hwang et al.16) reported that 58.2% of residents in long-term care facilities had received at least one PIM according to the 2012 Beers criteria and that the most prevalent PIMs were quetiapine, chlorpheniramine, risperidone, and zolpidem. A recent Korean retrospective study suggested that patients administered PIMs (defined using the 2012 Beers criteria) were more likely to be admitted to the hospital or visit emergency departments (ED).17) In that study, the most commonly used PIMs were chlorpheniramine, diazepam, metoclopramide, and diclofenac. Although most Korean studies examining PIMs have used the Beers criteria, 38 of 111 drugs (34.2%) considered PIMs according to the 2015 Beers criteria are unavailable in Korea. Due to the absence of a Korean PIMs list, it is hard for physicians to apply a PIMs screening tool in their daily practice. The 2010 Kim’s list has been the only published PIMs list for Korean older adults until now. However, it lacked a description of the applicable conditions or exceptions for each item and included some out-of-date items such as beta-blockers for heart failure and unavailable drugs such as tacrine, dipyridamole, and disopyramide.9) Therefore, an updated list was required to incorporate recent advances in this field.
As there is no official geriatric training system in Korea, many Korean doctors might be unfamiliar with PIMs for older adults. For this reason, the expert panel agreed to provide a concise rather than exhaustive list, which can be easily adopted by doctors who treat older people, including primary care physicians and surgeons. To reduce the number of PIMs items, the research steering team excluded items with CSS <9 points, with the possibility of re-entry in further Delphi rounds. We also merged some conditions sharing similar PIMs; for example, dementia with delirium, and history of falls with syncope and postural hypotension. Finally, we were able to produce a concise list including 62 general PIMs and 48 PIMs for 18 specific conditions compared to the 2015 Beers criteria, which included 113 general PIMs, 68 PIMs for 12 specific conditions, and 16 PIMs with caution. Despite the small number of items, the new list addresses common ADEs in the older population such as falls and cognitive problems,18) and also includes most of the prevalent PIMs in Korea.16,17) To reflect the fact that gastrointestinal bleeding is a major ADE causing hospitalization or ED visit,4,5) the panel included aspirin in older adults aged ≥80 years for cardiovascular primary prevention and aspirin plus clopidogrel for secondary stroke prevention in the list. The panel also suggested less stringent glycated hemoglobin targets (<7.5% or higher) for older patients with diabetes as a comment to reduce hypoglycemic events, which is another major cause of hospitalization related to ADEs.19)
Some panelists were skeptical about the inclusion of antipsychotics in this list because nonpharmacological options such as behavioral intervention for Behavioral and Psychological Symptoms of Dementia management are rarely available in Korean hospitals and long-term care facilities. However, with increasing evidence of harm associated with antipsychotics20) and conflicting evidence on their effectiveness,21) the expert panel agreed that antipsychotics should be included in the list with the comments, “Short-term and low-dose antipsychotic (such as haloperidol, risperidone, and quetiapine) use might be appropriate for delirium or dementia if nonpharmacological options (e.g., behavioral interventions) have failed or are not possible and the older adult is threatening substantial harm to self or others.” Apart from this list, the multifaceted effort to reduce antipsychotics use especially in a long-term care setting is urgently required in Korea. There was substantial concern regarding the use of over-the-counter drugs containing first-generation antihistamines and herbal medicine in older adults, but the panel excluded these items from the final list because they could have too wide and vague range of application without concrete evidence. The panel did not reach a consensus regarding megestrol until the final round of survey and it was, thus, excluded.
This study has several limitations. Because of limited resources, we generated the preliminary list without evidence review for each item but rather from the review of existing criteria, which could result in the omission of recently introduced items. However, in view of the limited number of methodologically high-quality studies of older patients, the Delphi method based on existing criteria has been acknowledged as an acceptable way to generate a domestic PIMs list.22,23) Next, the relatively small number and low diversity of panel members was another limitation of the study; therefore, the additional participation of cardiologists, neurologists, and more primary care physicians and clinical pharmacists is required in future updates of the list. In addition, future updates could consider drug-drug interactions and PIMs based on kidney function that were not addressed in this study to make the lists concise. Finally, there could be several exceptional clinical situations in which the drugs on this list should be prescribed for older patients. The expert panel tried to address this issue in the Delphi rounds and described typical exceptional situations for each item in comments, but they are not enough to cover diverse clinical settings. Therefore, this PIMs list is only a reference to support better clinical decisions in the prescription for older patients and cannot substitute for clinical decision-making in specific medical situations.
Computerized decision support, where electronic alerts are produced to guide the prescriber, is an effective intervention in improving medication prescription in older adults.24) In Korea, the nationwide drug utilization review (DUR) system was introduced in 2010 and the system is providing real-time safety alerts for prescribing long-acting benzodiazepines and TCA to older adults in outpatient clinics in Korea.25) It would be valuable to incorporate selected drugs from this list in the DUR after proper validation.
We hope that this explicit PIMs list helps Korean physicians to make individualized therapeutic decisions for their older patients and improve medication safety. Further prospective studies to validate its usefulness in various clinical setting and regular updating of the list are required.
ACKNOWLEDGMENTS
The authors thank Rho SY (Seoul Women’s University, Seoul) for advice regarding the Delphi process; Hwang HJ (Catholic Kwandong University College of Medicine, Incheon), Park YM (National Health Insurance Service Ilsan Hospital, Ilsan), Rho YK (Hallym University Medical Center, Seoul), Lee EJ (Asan Medical Center, Seoul), Kim ST (Seocho Cham Convalescence Hospital, Seoul), Lee HW (Seoul Medical Center, Seoul), and Park SH (Seoul Medical Center, Seoul) for their participation in the Delphi process; the Korean Geriatric Society, the Korean Academy of Clinical Geriatrics, the Korean Academy of Family Medicine, the Korean Association for Geriatric Psychiatry, and the Korean Association of Geriatric Hospitals for expert nominations. This study was supported by an internal research grant from the Seoul Medical Center (16-C11).
Notes
The researchers claim no conflicts of interest.