The Relationship Between Serum 25-Hydroxyvitamin D Levels and Physical Performance in Community-Dwelling Older Adults
Article information
Abstract
Background
Vitamin D deficiency is a risk factor for musculoskeletal health in older adults. While many studies have explored the relationship between vitamin D deficiency and fractures, few have examined the relationship between vitamin D and physical performance. We, therefore, sought to evaluate the relationship between serum 25-hydroxyvitamin D [25(OH)D] levels and physical performance in community-dwelling older adults.
Methods
A single-center, cross-sectional study was performed using data collected from 132 individuals aged 60 years or older who were living independently and who participated in the National Health Insurance Service health check-up between May and December 2016. Physical performance was assessed using a short physical performance battery (SPPB). Linear regression was used to examine the association between 25(OH)D levels and physical performance after adjustment for sociodemographic variables, behavioral characteristics, and body mass index.
Results
Approximately 36.5% of male and 50.7% of females had vitamin D levels indicative of deficiency (serum 25(OH)D <20.0 ng/mL). 25(OH)D levels were significantly associated with the chair stand SPPB scores in male and females but not with those of the walking and balance tests. After adjustment for potential confounders, there remained a linear association between 25(OH)D levels and the chair stand test for both sexes.
Conclusion
Lower serum 25(OH)D levels were associated with poor physical performance (chair stand score) among community-dwelling older adults in Korea. Further prospective studies are needed to verify these results.
INTRODUCTION
Vitamin D plays an important role in bone and mineral metabolism. Severe vitamin D deficiency leads to bone diseases such as rickets in children and osteomalacia in adults.1) Humans acquire vitamin D in two ways: from the diet and via cutaneous synthesis driven by sunlight. The active form of vitamin D is a potent steroid hormone that promotes the proliferation and differentiation of muscle cells via vitamin D receptors (VDRs) in skeletal muscle.2–4) Therefore, inadequate vitamin D can lead to the disturbance of muscle protein synthesis and reduced muscle mass and strength.5–8) The increased secretion of parathyroid hormone in vitamin D insufficiency (secondary hyperparathyroidism) contributes to bone fragility, fractures, and impaired muscle function.1,9,10)
Vitamin D deficiency has recently attracted increasing interest among geriatric clinicians because the condition is common in older adults and can result in increased susceptibility to falls and osteoporotic fractures later in life.6,11) In a recent Korean study, the prevalence of inadequate vitamin D levels was high among older urban populations, at 63.6% in males and 63.7% in females.12) In Korea, where a majority of the population lives in the cities, senior citizens spend significant time indoors and have insufficient ultraviolet B exposure for adequate cutaneous vitamin D production. Even when they go outdoors, older adults tend to apply sunscreen and avoid wearing short-sleeved shirts or short shorts. Furthermore, the capacity to synthesize vitamin D in the skin decreases with age.13) The number of VDRs in skeletal muscle also decreases with age, which can lead to a decline in muscle strength in older populations.14)
Although the effect of vitamin D in maintaining musculoskeletal health is well established, the understanding of how vitamin D affects physical performance remains limited.6,11,14–18) Previous studies have explored the relationship between vitamin D deficiency and fracture but relatively few have focused on the relationship between vitamin D and physical performance in Asian populations, especially in Korean older populations.19) Therefore, the aim of this study was to examine the association between vitamin D levels and physical performance among community-dwelling older adults in Korea.
MATERIALS AND METHODS
Study Population
This single-center, cross-sectional study was conducted using data collected from 165 individuals aged 60 years or older who participated in the National Health Insurance Service health check-ups between May 10, 2016, and December 8, 2016. The inclusion criteria were as follows: (1) age 60 years or over; (2) able to walk 10 m independently with or without a walking aid; and (3) living independently and able to go outside on a regular basis. The exclusion criteria were as follows: (1) withdrawal of consent; (2) incomplete data; (3) prolonged glucocorticoid use (more than 3 months at any time); (4) hormone replacement therapy; (5) use of calcium or vitamin D as a nutritional supplement; (6) use of osteoporosis medication or advanced renal failure (estimated glomerular filtration rate <30 mL/min); (7) cognitive impairments limiting the ability to understand the motor tasks required during testing; (8) central nervous system disorders, peripheral nervous system disorders, or myopathies; (9) previous lower extremity surgery, and (10) current or past cancer.
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. Ethical approval was provided by the Institutional Review Board of Kyungpook National University Hospital (IRB protocol number KNUH 2016-04-016-011). All participants were informed about the study and provided written consent to participate.
Demographic and Clinical Characteristics
Height, weight, and waist circumference were measured by a trained nurse. Waist circumference was measured as the smallest circumference at or below the costal margin, while hip circumference was measured at the level of the greater trochanter using a fiberglass tape measure at a tension of 600 g. Body mass index (BMI) was calculated as weight in kilograms divided by height in square meters (kg/m2). Obesity was defined as a BMI ≥25 kg/m2.20) After 5 minutes of rest, blood pressure was measured twice using automatic oscillometric devices (BPBIO320; InBody, Seoul, Korea) and the mean value (mmHg) was used as the final measurement.
To collect detailed information regarding smoking status, alcohol consumption, and physical activity, standardized questionnaires were administered by trained health professionals. Cigarette smoking was assessed using two questions validated by the Centers for Disease Control and Prevention Behavioral Risk Factor Surveillance System: “Have you smoked at least 100 cigarettes in your entire life?” and “Do you now smoke cigarettes every day, some days, or not at all?” The responses were grouped into two categories: never smoker and ever smoker. Ever smokers were defined as those persons who reported smoking at least 100 cigarettes in their lifetime. Never smokers were defined as persons who reported never having smoked 100 cigarettes.21) Self-reported alcohol consumption was assessed as the number of drinks/week (one drink=14 g of alcohol). Based on their responses, the participants were categorized as social or binge drinkers. As per the Center for Disease Control and Prevention criteria, binge drinking was defined as the consumption of more than five and four (for males and females, respectively) consecutive drinks of any type of alcohol within the past 12 months.22) Participants were asked a binary (yes/no) question related to physical activity. People who reported moderate physical activity (3–6 metabolic equivalents) for at least 150 minutes per week were defined as having “good physical activity”; the remainder were defined as having “poor physical activity”.23)
Comorbidity was defined as being diagnosed with three or more of the following diseases: stroke, heart disease (myocardial infarction or angina), hypertension, diabetes mellitus, dyslipidemia, or pulmonary tuberculosis.
Measurement of Vitamin D and Other Laboratory Tests
After fasting for at least 8 hours, all participants visited the health-promotion centers on the morning of the appointed day. Serum was isolated by centrifugation (F5220; Kubota Corporation, Saitama, Japan) within 1 hour of collection and kept refrigerated (2°C–8°C) until use. The serum concentration of 25(OH)D was used as a measure of vitamin D status. Levels of 25(OH)D were measured by radioimmunoassay using a Packard COBRA Gamma Counter (DiaSorin; Saluggia, Vercelli, Italy). The reference range for the assay is 8.9–37.6 ng/mL, with intra- and inter-assay coefficients of variation of 8.1% and 10.2%, respectively. Vitamin D deficiency was defined as a 25(OH)D level <20 ng/mL, and the definitions of insufficiency was defined as a 25(OH)D level <30 ng/mL.24) The following were measured in all participants: markers of liver function (alanine aminotransferase and aspartate aminotransferase); lipid profiles (total cholesterol, low-density lipoprotein, high-density lipoprotein, and triglycerides); complete blood count; blood urea nitrogen; serum creatinine; and fasting blood glucose. Serum creatinine was measured by a Beckman Coulter analyzer (AU 2700; Beckman Coulter Diagnostics, Brea, CA, USA) using Jaffe’s method. Calibration was performed using isotope dilution mass spectroscopy. Serum total cholesterol, low-density lipoprotein, high-density lipoprotein, triglycerides, and glucose, as well as the other basic biochemical blood parameters, were measured using standard chemical and enzymatic commercial methods on a Hitachi analyzer (D-2400; Hitachi High-Technologies Corporation, Tokyo, Japan).
Assessment of Physical Performance
A short physical performance battery (SPPB) was used to assess physical performance. The SPPB test is an objective assessment tool for the evaluation of lower extremity function in older adults. It was developed by the National Institute of Aging and is used widely in the geriatric literature.25) The SPPB includes a 3-meter walking test, a chair stand test (standing up from a chair as quickly as possible without using hands (repeated five times), and a balance test (standing with feet in “tandem stand” for up to 30 seconds). Participants score 0–4 points for each test, with a maximum of 12 points and higher scores indicating better performance. Scores of 0–3, 4–6, 7–9, and 10–12 are classified as severe, moderate, mild, and minimal lower extremity limitations, respectively.26)
Statistical Analyses
Statistical calculations were performed using IBM SPSS statistics for Windows, version 21.0 (IBM Co., Armonk, NY, USA). P-values of less than 0.05 were considered statistically significant. T-tests or Kruskal–Wallis tests were used to compare continuous variables, while Pearson’s chi-square tests were used to compare categorical variables. The summarized statistics are presented as means±standard deviation (continuous variables) and as numbers (percentage) (categorical variables). The participants were divided into quartiles according to their 25(OH) D level. Since serum vitamin D levels are sex-dependent, males and female older adults were subjected to stratified analyses.27–29) Separate analyses were performed for each physical function outcome test (3-meter walking test, chair stand test, and balance test). In Model 1, linear regression analysis was performed with serum 25(OH)D as an independent variable and each SPPB score as a dependent variable. The model was adjusted for age, smoking status, physical activity, and BMI (all factors that might affect physical performance).30–32) Model 2 was adjusted for age. Model 3 was adjusted for the variables used in Model 2 plus smoking status, alcohol consumption, physical activity, BMI, and comorbidities. All independent variables were included in the regression model simultaneously using the enter method.
RESULTS
Subject Characteristics
Overall, 165 participants who met the inclusion criteria were recruited; 33 participants who withdrew their consent were excluded. Complete data were available for 132 (63 males, 47.7%) participants who were enrolled in the final analysis. All participants were Korean. The sex-specific characteristics of the study population are shown in Table 1. The mean ages of the males and females in the study were 68.30 (range, 60–85) and 68.45 (range, 60–84) years, respectively.
Obesity was more common in males than in females (41.3% vs. 27.5%; p=0.070). Significantly fewer females than males were ever smokers (1.4% vs. 12.7%; p<0.001). Binge drinking was reported by 11.1% of males and by no females (p=0.004). In addition, 30.2% of males and 18.8% of females were physically active and 15.9% of males and 10.1% of females had comorbidities.
Vitamin D Levels
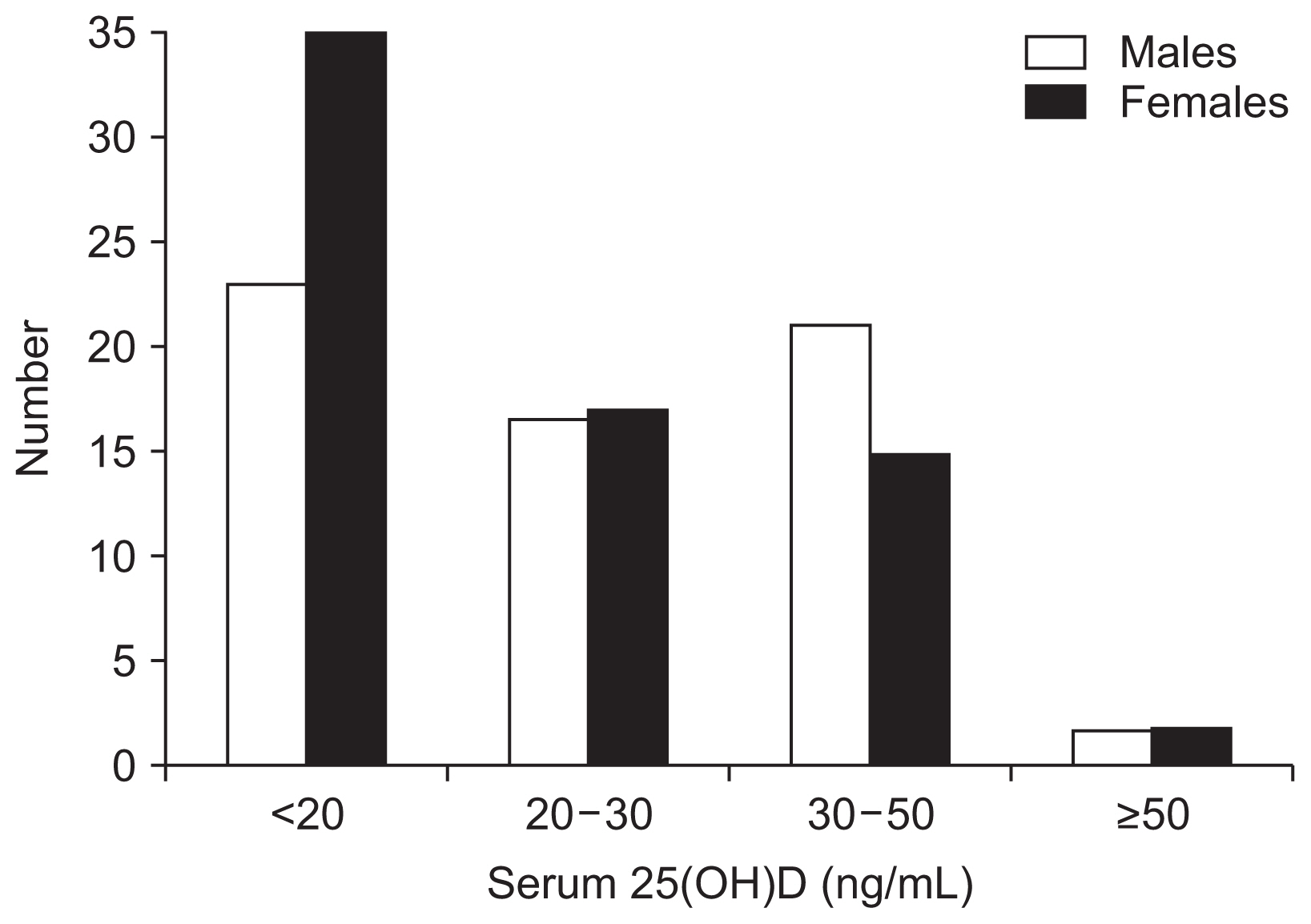
Fig. 1 shows the distribution of 25(OH)D levels. Vitamin D deficiency (serum 25(OH)D, <20.0 ng/mL) was highly prevalent in this population. More females than males were deficient (36.5% and 50.7%, respectively). There were no significant differences in mean serum 25(OH)D levels between males (23.61±9.07 ng/mL) and females (22.55±11.33 ng/mL) (p=0.556).
Physical Performance According to Vitamin D Quartile
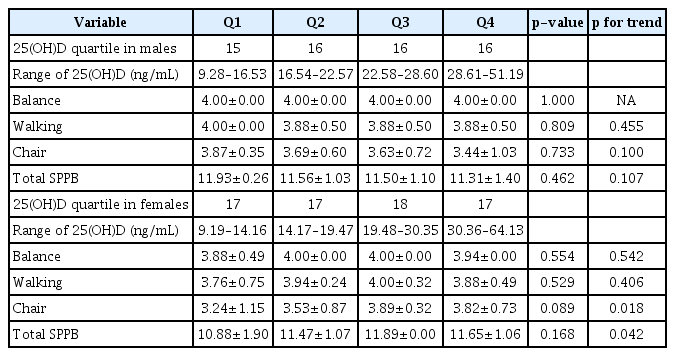
The results of the physical performance tests according to serum 25(OH)D quartile are shown in Table 2. For females, 25(OH)D status was positively associated with better chair stand test scores (p for trend=0.018) but there were no significant differences among the 25(OH)D quartiles with respect to the other SPPB scores. For males, the 25(OH) D status across all four quartiles was positively associated with balance scores and there were no significant differences among 25(OH)D quartiles with respect to the other SPPB scores.
Associations Between Vitamin D Levels and Physical Performance
Model 1 revealed that lower 25(OH)D levels in females were associated with worse chair stand test scores (β=0.213, p=0.018), independent of age, BMI, comorbidity, and lifestyle factors (alcohol consumption, smoking, and physical activity) (β=0.176, p=0.046; Model 3). However, there were no associations between 25(OH)D levels and balance test and walking test scores (p=0.626 and p=0.615, respectively; Model 3).
For males, Models 1 and 2 revealed no significant association between 25(OH)D levels and any SPPB score. However, in the fully-adjusted model (Model 3), lower 25(OH) D levels were significantly associated with a worse chair stand score (β=−0.168, p=0.036) (Table 3).
DISCUSSION
The results of this study showed that vitamin D insufficiency was prevalent among community-dwelling older individuals in Korea. Most of the subjects in this study (77.3%) had 25(OH)D levels below 30 ng/mL. These results are similar to those reported by a previous study in Korea showing that 79.3% of males and 75.3% of females had vitamin D levels below 30 ng/mL.33) However, previous studies suggest that the prevalence of inadequate vitamin D levels in other countries is not as high as that in Korea. For example, the prevalence of vitamin D deficiency in Japan was much lower, at only around 5% of active older persons. In the present study, 36.51% of males and 50.72% of females had vitamin D levels below 20 ng/mL; however, a recent study of Chinese individuals aged 60 years or older also reported that 34.1% of males and 44.0% of females had inadequate vitamin D levels (<20 ng/mL).34) Furthermore, countries with low latitudes, such as Australia, Indonesia, and Brazil, also have a lower prevalence of vitamin D deficiency.13) These differences might be due to the differences in age, ethnicity, lifestyle, diets fortified with vitamin D, or geographical difference leading to differences in sun exposure.35,36)
We observed an association between low serum 25(OH) D levels and the chair stand scores in both sexes after adjustment for confounding factors (Model 3). However, there was no significant association with the other two parameters of the SPPB. There are two possible explanations for the discrepancy among SPPB parameters. First, the chair stand test requires more proximal lower extremity muscle strength.37) Second, the differences may be due to the characteristics of the study population. This study only included relatively healthy older adults who went outdoors regularly and lived independently with or without the use of walking aids.
In this study, vitamin D insufficiency affected males and females differently. This difference between males and females could be explained by sex hormones and VDR gene polymorphisms. Sex hormones play a role in regulating vitamin D and calcium homeostasis. Estrogen regulates VDR expression and 1a-hydroxylase activity, while testosterone stimulates the expression of intestinal calcium channels.38,39) The fact that males tend to be more physically active than females might also influence the results. In the present study, 30.2% of males but only 18.8% of females exercised for more than 150 minutes per week. Females with poor SPPB scores who spend little time outdoors are likely to have low 25(OH)D levels, which is consistent with results of previous studies.40,41)
The other possible explanations for why SPPB scores other than the chair stand score were not to related to vitamin D levels include age-related reductions in VDR expression.14) Low vitamin D levels are common in older adults and long-term vitamin D deficiency might lead to the down-regulation of VDR.27) In addition, VDR polymorphisms may also result in variable susceptibility to age-related reductions in muscle performance.31) Although we used a validated tool (SPPB) to evaluate physical performance, it may not fully reflect the overall physical performance.
The findings presented in this study are consistent with those of previous cross-sectional studies. For example, recent studies reported that higher 25(OH)D levels are associated with better musculoskeletal function in the lower extremities.42) In the present study, the fully-adjusted model revealed a positive association between serum 25(OH) D levels and the chair stand scores in both sexes. Lower extremity function is considered an important factor for musculoskeletal health in older adults, as improved muscle strength can prevent disability, falls, and fractures.18) Our results provide additional evidence that 25(OH)D is associated with physical function.
This study has several limitations. The first is a possible selection bias due to the recruitment of subjects who were in good physical condition. Our use of volunteers might mean that we inadvertently enrolled a relatively healthy older population because they were not inpatients or nursing home residents. Also, we excluded people who are not able to walk 10 m independently. Second, this was a cross-sectional study; as such, we cannot determine cause-effect relationships, which may be bidirectional. Low vitamin D levels may cause poor physical performance. Conversely, physically active subjects are likely to spend time outdoors and longer sunlight exposure leads to higher endogenous vitamin D synthesis than that in sedentary subjects. Finally, the small sample size meant that the study lacked sufficient power to discriminate between subgroups. Individuals with lower 25(OH)D levels might be reasonably expected to have a more severe impairment with respect to SPPB; however, we could not perform subgroup analysis of patients with respect to SPPB scores or different vitamin D levels.
This study also has several strengths. First, we used the SPPB test, which is a standardized and validated test for use in older populations that is a good predictor of falls, inflammatory reactions, and declining function; it also assesses maximum functional outcome rather than lower extremity disability.18,25) Second, the high prevalence of 25(OH)D deficiency in this population made it a good sample in which to investigate the relationship between vitamin D and physical performance.38) All of the subjects lived in an urban area in Korea, which is likely related to a sun-shy lifestyle and poor exposure to sunlight when compared to sunnier countries such as Australia, Indonesia, and Brazil.13) Third, we used an easily replicable model. The strategy used in this study only added two tests (serum 25(OH)D levels and SPPB) to the routine laboratory tests commonly used in outpatient clinics or during regular check-ups. Therefore, it is easy to implement, even in a primary clinic or in hospital-based care, because training or specialized equipment are not required.
Lower serum 25(OH)D levels were associated with poor chair stand scores in ambulatory Koreans aged over 60 years, independent of age, BMI, smoking status, physical activity, and comorbidities. There was no association between serum 25(OH)D levels and the other SPPB scores. Serum 25(OH)D may be an important indicator for various health outcomes, including physical performance, in older adults. Further longitudinal studies are needed to clarify the effects of vitamin D supplementation with respect to improving skeletal muscle function and reducing falls and fractures. In addition, vitamin D levels in older adults should be monitored. Patients should be educated about the importance of sun exposure along with a healthy lifestyle. Primary prevention of vitamin D deficiency is needed to improve musculoskeletal health in later life. Public health strategies should target this population.
ACKNOWLEDGMENTS
This work was funded by Biomedical Research Institute grant, Kyungpook National University Hospital (2016-NEW-06).
Notes
The researchers claim no conflicts of interest.